Papers
Lo resaltado
(Para una lista completa, revisa abajo)
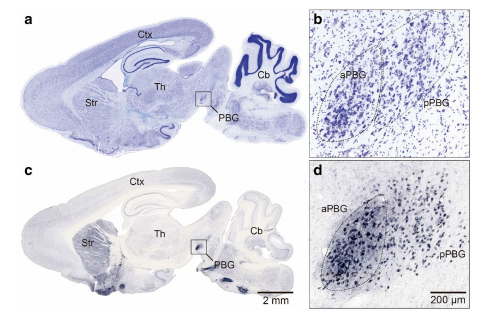
Abstract. The parabigeminal nucleus (PBG) is the mammalian homologue to the isthmic complex of other vertebrates. Optogenetic stimulation of the PBG induces freezing and escape in mice, a result thought to be caused by a PBG projection to the central nucleus of the amygdala. However, the isthmic complex, including the PBG, has been classically considered satellite nuclei of the Superior Colliculus (SC), which upon stimulation of its medial part also triggers fear and avoidance reactions. As the PBG-SC connectivity is not well characterized, we investigated whether the topology of the PBG projection to the SC could be related to the behavioral consequences of PBG stimulation. To that end, we performed immunohistochemistry, in situ hybridization and neural tracer injections in the SC and PBG in a diurnal rodent, the Octodon degus. We found that all PBG neurons expressed both glutamatergic and cholinergic markers and were distributed in clearly defined anterior (aPBG) and posterior (pPBG) subdivisions. The pPBG is connected reciprocally and topographically to the ipsilateral SC, whereas the aPBG receives afferent axons from the ipsilateral SC and projected exclusively to the contralateral SC. This contralateral projection forms a dense field of terminals that is restricted to the medial SC, in correspondence with the SC representation of the aerial binocular field which, we also found, in O. degus prompted escape reactions upon looming stimulation. Therefore, this specialized topography allows binocular interactions in the SC region controlling responses to aerial predators, suggesting a link between the mechanisms by which the SC and PBG produce defensive behaviors.
Deichler, A., Carrasco, D., Lopez-Jury, L., Vega-Zuniga, T., Márquez, N., Mpodozis, J., & Marín, G. J. (2020)
Scientific Reports, 10, 16220: 1-16. doi: 10.1038/s41598-020-72848-0.
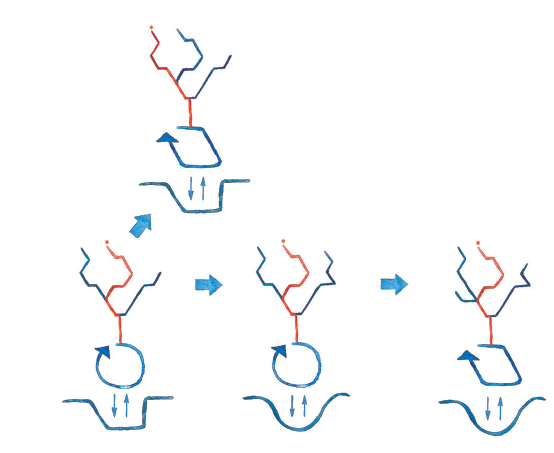
Abstract. Living beings are autopoietic systems with highly context‐dependent structural dynamics and interactions, that determine whether a disturbance in the genotype or environment will lead or not to phenotypic change. The concept of epigenesis entails how a change in the phenotype may not correspond to a change in the structure of an earlier developmental stage, including the genome. Disturbances of embryonic structure may fail to change the phenotype, as in regulated development, or when different genotypes are associated to a single phenotype. Likewise, the same genotype or early embryonic structure may develop different phenotypes, as in phenotypic plasticity. Disturbances that fail to trigger phenotypic change are considered neutral, but even so, they can alter unexpressed developmental potential. Here, we present conceptual diagrams of the “epigenic field”: similar to Waddington’s epigenetic landscapes, but including the ontogenic niche (organism/environment interactional dynamics during ontogeny) as a factor in defining epigenic fields, rather than just selecting among possible pathways. Our diagrams illustrate transgenerational changes of genotype, ontogenic niche, and their correspondence (or lack thereof) with changes of phenotype. Epigenic fields provide a simple way to understand developmental constraints on evolution, for instance: how constraints evolve as a result of developmental system drift; how neutral changes can be involved in genetic assimilation and de‐assimilation; and how constraints can evolve as a result of neutral changes in the ontogenic niche (not only the genotype). We argue that evolutionary thinking can benefit from a framework for evolution with conceptual foundations at the organismal level.
Vargas, A. O., Botelho, J. F. & Mpodozis, J. (2020)
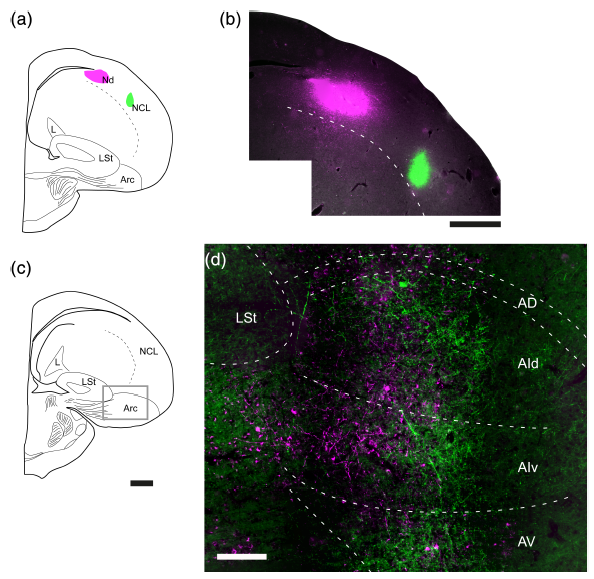
Abstract. The sensory–motor division of the avian arcopallium receives parallel inputs from primary and high‐order pallial areas of sensory and vocal control pathways, and sends a prominent descending projection to ascending and premotor, subpallial stages of these pathways. While this organization is well established for the auditory and trigeminal systems, the arcopallial subdivision related to the tectofugal visual system and its descending projection to the optic tectum (TeO) has been less investigated. In this study, we charted the arcopallial area displaying tectofugal visual responses and by injecting neural tracers, we traced its connectional anatomy. We found visual motion‐sensitive responses in a central region of the dorsal (AD) and intermediate (AI) arcopallium, in between previously described auditory and trigeminal zones. Blocking the ascending tectofugal sensory output, canceled these visual responses in the arcopallium, verifying their tectofugal origin. Injecting PHA‐L into the visual, but not into the auditory AI, revealed a massive projection to tectal layer 13 and other tectal related areas, sparing auditory, and trigeminal ones. Conversely, CTB injections restricted to TeO retrogradely labeled neurons confined to the visual AI. These results show that the AI zone receiving tectofugal inputs sends top‐down modulations specifically directed to tectal targets, just like the auditory and trigeminal AI zones project back to their respective subpallial sensory and premotor areas, as found by previous studies. Therefore, the arcopallium seems to be organized in a parallel fashion, such that in spite of expected cross‐modal integration, the different sensory–motor loops run through separate subdivisions of this structure.
Fernández, M., Morales, C., Durán, E., Fernández-Colleman, S., Sentis, E., Mpodozis, J., Karten, H. J., & Marín, G. J. (2020)
Journal of Comparative Neurology, 528: 597-623. doi: 10.1002/cne.24775.
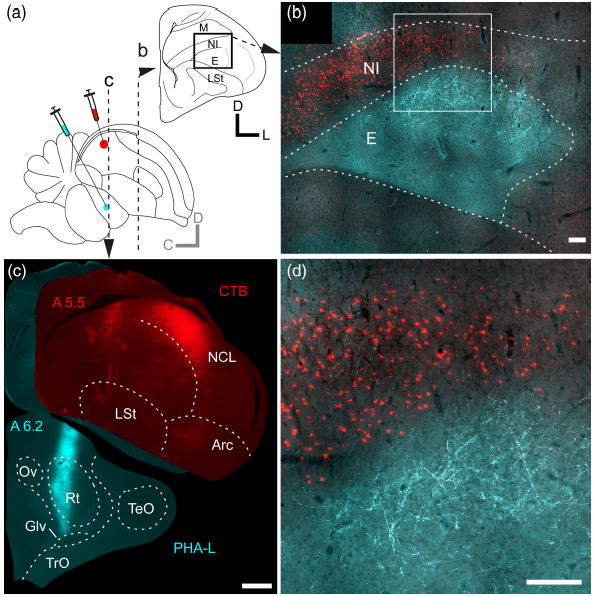
Abstract. Recent reports have shown that the avian visual dorsal ventricular ridge (DVR) is organized as a trilayered complex, in which the forming layers—the thalamo‐recipient entopallium (E), an overlaying nidopallial stripe called intermediate nidopallium (NI), and the dorsally adjacent mesopallium ventrale—appear to be extensively interconnected by topographically organized columns of reciprocal axonal processes running perpendicular to the layers, an arrangement highly reminiscent to that of the sensory cortices of mammals. In the present report, we implemented in vivo anterograde and retrograde tracing techniques aiming to elucidate the organization of the connections of this complex with other pallial areas. Previous studies have shown that the efferent projections of the visual DVR originate mainly from the NI and E, reaching several distinct associative and premotor nidopallial areas. We found that the efferents from the visual DVR originated solely from the NI, and confirmed that the targets of these projections were the pallial areas described by previous studies. We also found novel projections from the NI to the visual hyperpallium, and to the lateral striatum. Moreover, we found that these projections were reciprocal, topographically organized, and originated from different cell populations within the NI. We conclude that the NI constitutes a specialized layer of the visual DVR that form the core of a dense network of highly specific connections between this region and other higher order areas of the avian pallium. Finally, we discuss to what extent these hodological properties resemble those of the mammalian cortical layers II/III.
Fernández, M., Ahumada-Galleguillos, P., Sentis, E., Marín, G., & Mpodozis, J. (2020)
Journal of Comparative Neurology, 528: 321– 359. doi: 10.1002/cne.24757.
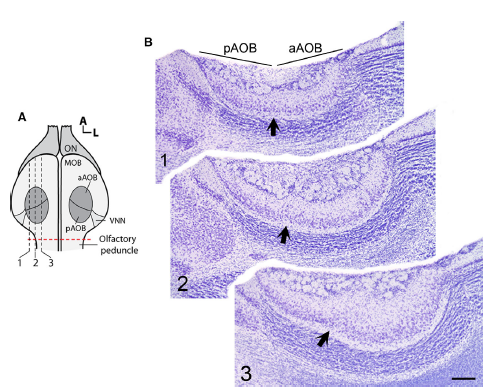
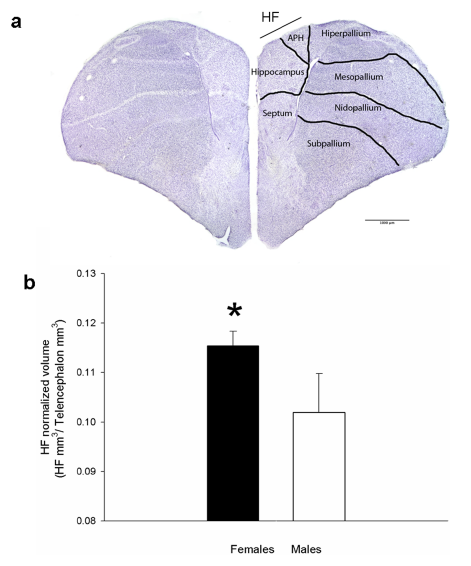
Abstract. In mammals, the accessory olfactory or vomeronasal system exhibits a wide variety of anatomical arrangements. In caviomorph rodents, the accessory olfactory bulb (AOB) exhibits a dichotomic conformation, in which two subdomains, the anterior (aAOB) and the posterior (pAOB), can be readily distinguished. Interestingly, different species of this group exhibit bias of different sign between the AOB subdomains (aAOB larger than pAOB or vice versa). Such species‐specific biases have been related with contrasting differences in the habitat of the different species (e.g. arid vs. humid environments). Aiming to deepen these observations, we performed a morphometric comparison of the AOB subdomains between two sister species of octodontid rodents, Octodon lunatus and Octodon degus. These species are interesting for comparative purposes, as they inhabit similar landscapes but exhibit contrasting social habits. Previous reports have shown that O. degus, a highly social species, exhibits a greatly asymmetric AOB, in which the aAOB has twice the size of the pAOB and features more and larger glomeruli in its glomerular layer (GL). We found that the same as in O. degus, the far less social O. lunatus also exhibits a bias, albeit less pronounced, to a larger aAOB. In both species, this bias was also evident for the mitral/tufted cells number. But unlike in O. degus, in O. lunatus this bias was not present at the GL. In comparison with O. degus, in O. lunatus the aAOB GL was significantly reduced in volume, while the pAOB GL displayed a similar volume. We conclude that these sister species exhibit a very sharp difference in the anatomical conformation of the AOB, namely, the relative size of the GL of the aAOB subdomain, which is larger in O. degus than in O. lunatus. We discuss these results in the context of the differences in the lifestyle of these species, highlighting the differences in social behaviour as a possible factor driving to distinct AOB morphometries.
Fernández-Aburto, P., Delgado, S. E., Sobrero, R., & Mpodozis, J. (2020)
Journal of Anatomy, 236: 612-621. doi: 10.1111/joa.13126.
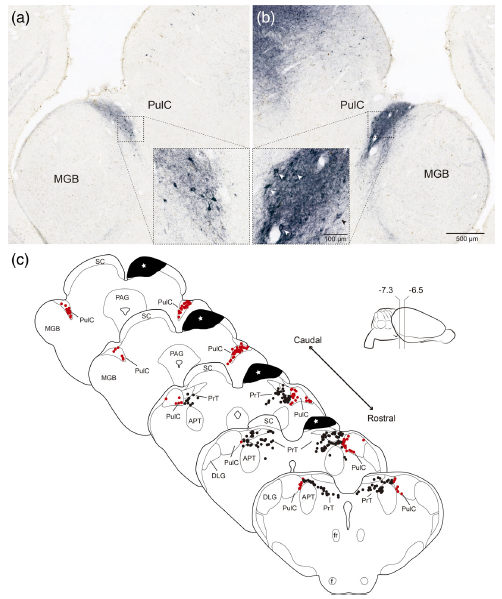
Abstract. A defining feature of the amniote tecto‐fugal visual pathway is a massive bilateral projection to the thalamus originating from a distinct neuronal population, tectal ganglion cells (TGCs), of the optic tectum/superior colliculus (TeO/SC). In sauropsids, the thalamic target of the tecto‐fugal pathway is the nucleus rotundus thalami (Rt). TGCs axons collateralize en route to Rt to target the nucleus pretectalis principalis (PT), which in turn gives rise to bilateral projection to the TeO. In rodents, the thalamic target of these TGCs afferents is the caudal division of the pulvinar complex (PulC). No pretectal structures in receipt of TGC collaterals have been described in this group. However, Baldwin et al. (Journal of Comparative Neurology, 2011;519(6):1071–1094) reported in the squirrel a feedback projection from the PulC to the SC. Pulvino‐tectal (Pul‐T) cells lie at the caudal pole of the PulC, intermingled with the axonal terminals of TGCs. Here, by performing a combination of neuronal tracing, immunohistochemistry, immunofluorescence, and in situ hybridization, we characterized the pattern of projections, neurochemical profile, and genoarchitecture of Pul‐T cells in the diurnal Chilean rodent Octodon degus. We found that Pul‐T neurons exhibit pretectal, but not thalamic, genoarchitectonical markers, as well as hodological and neurochemical properties that match specifically those of the avian nucleus PT. Thus, we propose that Pul‐T cells constitute a pretectal cell population hidden within the dorsal thalamus of mammals. Our results solve the oddity entailed by the apparent existence of a noncanonic descending sensory thalamic projection and further stress the conservative character of the tectofugal pathway.
Deichler, A., Carrasco, D., Gonzalez-Cabrera, C., Letelier, J. C., Marín, G., & Mpodozis, J. (2019)
Journal of Comparative Neurology, 527: 372– 391. doi: 10.1002/cne.24540.
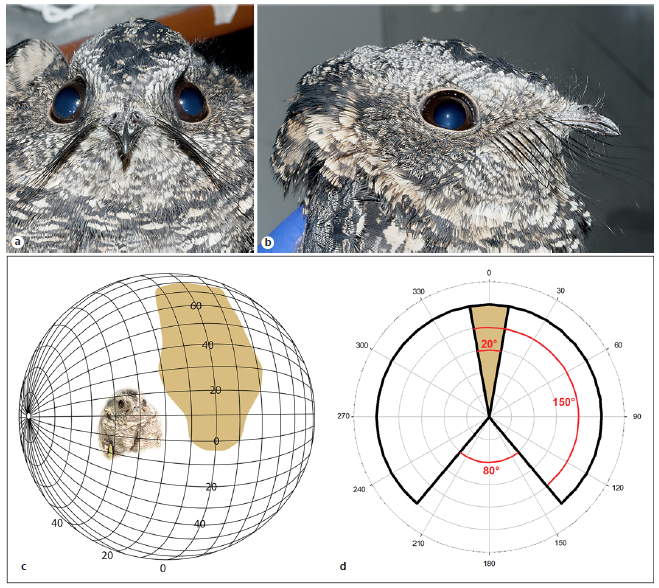
Abstract. Nocturnal animals that rely on their visual system for foraging, mating, and navigation usually exhibit specific traits associated with living in scotopic conditions. Most nocturnal birds have several visual specializations, such as enlarged eyes and an increased orbital convergence. However, the actual role of binocular vision in nocturnal foraging is still debated. Nightjars (Aves: Caprimulgidae) are predators that actively pursue and capture flying insects in crepuscular and nocturnal environments, mainly using a conspicuous “sit-and-wait” tactic on which pursuit begins with an insect flying over the bird that sits on the ground. In this study, we describe the visual system of the band-winged nightjar (Systellura longirostris), with emphasis on anatomical features previously described as relevant for nocturnal birds. Orbit convergence, determined by 3D scanning of the skull, was 73.28°. The visual field, determined by ophthalmoscopic reflex, exhibits an area of maximum binocular overlap of 42°, and it is dorsally oriented. The eyes showed a nocturnal-like normalized corneal aperture/axial length index. Retinal ganglion cells (RGCs) were relatively scant, and distributed in an unusual oblique-band pattern, with higher concentrations in the ventrotemporal quadrant. Together, these results indicate that the band-winged nightjar exhibits a retinal specialization associated with the binocular area of their dorsal visual field, a relevant area for pursuit triggering and prey attacks. The RGC distribution observed is unusual among birds, but similar to that of some visually dependent insectivorous bats, suggesting that those features might be convergent in relation to feeding strategies.
Salazar, J. E., Severin, D., Vega-Zuniga, T., Fernández-Aburto, P., Deichler, A., Sallaberry, M. A., & Mpodozis, J. (2019)
Brain, Behavior and Evolution, 94(suppl 1-4): 27-36. doi: 10.1159/000504162.
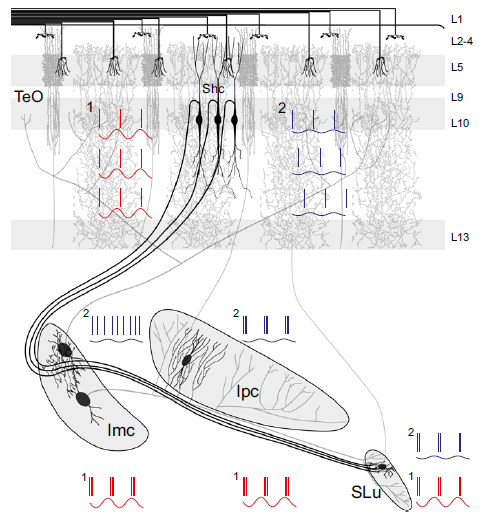
Abstract. The optic tectum (TeO), or superior colliculus, is a multisensory midbrain center that organizes spatially orienting responses to relevant stimuli. To define the stimulus with the highest priority at each moment, a network of reciprocal connections between the TeO and the isthmi promotes competition between concurrent tectal inputs. In the avian midbrain, the neurons mediating enhancement and suppression of tectal inputs are located in separate isthmic nuclei, facilitating the analysis of the neural processes that mediate competition. A specific subset of radial neurons in the intermediate tectal layers relay retinal inputs to the isthmi, but at present it is unclear whether separate neurons innervate individual nuclei or a single neural type sends a common input to several of them. In this study, we used in vitro neural tracing and cell-filling experiments in chickens to show that single neurons innervate, via axon collaterals, the three nuclei that comprise the isthmotectal network. This demonstrates that the input signals representing the strength of the incoming stimuli are simultaneously relayed to the mechanisms promoting both enhancement and suppression of the input signals. By performing in vivo recordings in anesthetized chicks, we also show that this common input generates synchrony between both antagonistic mechanisms, demonstrating that activity enhancement and suppression are closely coordinated. From a computational point of view, these results suggest that these tectal neurons constitute integrative nodes that combine inputs from different sources to drive in parallel several concurrent neural processes, each performing complementary functions within the network through different firing patterns and connectivity.
Garrido-Charad, F., Vega-Zuniga, T., Gutiérrez-Ibáñez, C., Fernandez, P., López-Jury, L., González-Cabrera, C., Karten, H. J., Luksch, H., & Marín, G. J. (2018)
Proceedings of the National Academy of Sciences, 115(32) E7615-E7623. doi: 10.1073/pnas.1804517115.
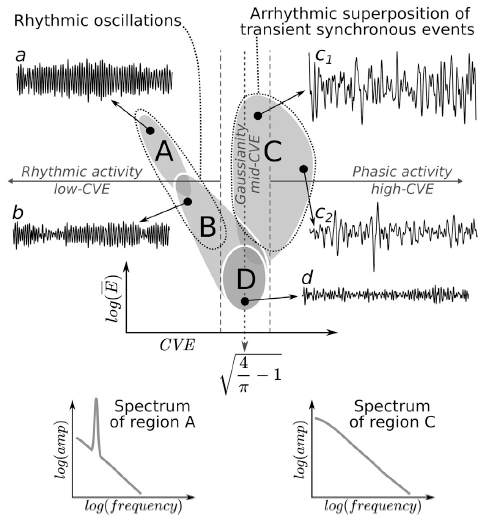
Abstract. Traditionally, EEG is understood as originating from the synchronous activation of neuronal populations that generate rhythmic oscillations in specific frequency bands. Recently, new neuronal dynamics regimes have been identified (e.g. neuronal avalanches) characterized by irregular or arrhythmic activity. In addition, it is starting to be acknowledged that broadband properties of EEG spectrum (following a 1/f law) are tightly linked to brain function. Nevertheless, there is still no theoretical framework accommodating the coexistence of these two EEG phenomenologies: rhythmic/narrowband and arrhythmic/broadband. To address this problem, we present a new framework for EEG analysis based on the relation between the Gaussianity and the envelope of a given signal. EEG Gaussianity is a relevant assessment because if EEG emerges from the superposition of uncorrelated sources, it should exhibit properties of a Gaussian process, otherwise, as in the case of neural synchronization, deviations from Gaussianity should be observed. We use analytical results demonstrating that the coefficient of variation of the envelope (CVE) of Gaussian noise (or any of its filtered sub-bands) is the constant sqrt(4π−1)≈0.523, thus enabling CVE to be a useful metric to assess EEG Gaussianity. Furthermore, a new and highly informative analysis space (envelope characterization space) is generated by combining the CVE and the envelope average amplitude. We use this space to analyze rat EEG recordings during sleep-wake cycles. Our results show that delta, theta and sigma bands approach Gaussianity at the lowest EEG amplitudes while exhibiting significant deviations at high EEG amplitudes. Deviations to low-CVE appeared prominently during REM sleep, associated with theta rhythm, a regime consistent with the dynamics shown by the synchronization of weakly coupled oscillators. On the other hand, deviations to high-CVE, appearing mostly during NREM sleep associated with EEG phasic activity and high-amplitude Gaussian waves, can be interpreted as the arrhythmic superposition of transient neural synchronization events. These two different manifestations of neural synchrony (low-CVE/high-CVE) explain the well-known spectral differences between REM and NREM sleep, while also illuminating the origin of the EEG 1/f spectrum.
Díaz, J., Bassi, A., Coolen, A., Vivaldi, E. A., & Letelier, J. C. (2018)
NeuroImage, 172: 575-585. doi: 10.1016/j.neuroimage.2018.01.063.

This special topic proposes an integrative view of brain evolution involving ecology, behavior, cognition and neurodevelopmental processes. We address three main questions, (i) the role of sensorimotor systems in brain evolution, (ii) the evolution of computational capacities and neural circuits, and (iii) the role of development in shaping brain evolution.
Aboitiz, F., Concha, M. L., González-Billault, C., & Mpodozis, J. (2018)
Frontiers in Neuroscience, 12, 447: 1-2. doi: 10.3389/fnins.2018.00447.

Functional approaches to systems, while of heuristic value in many cases, may lead to a neglect of some fundamental aspects of systemic phenomenology. The adoption of an alternative structural/mechanistic approach can be very enlightening to unveil, and effective to solve, the conceptual and heuristic limitations resulting from the adoption of the functional approach. The epistemic history of immunology, as described in the target article, offers compelling evidences of this situation.
Mpodozis, J. (2017)
Constructivist Foundations, 13(1): 147–149.
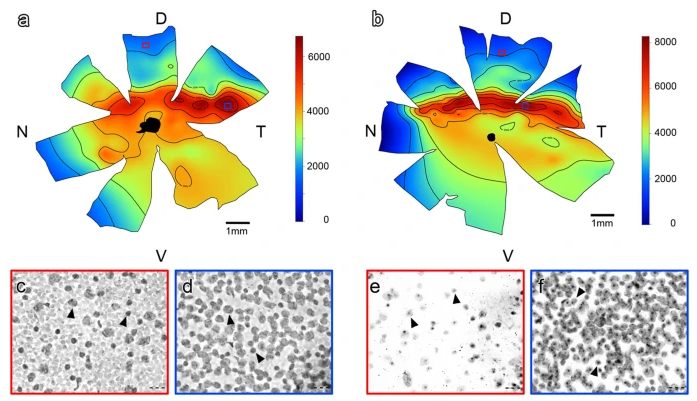
Abstract. To what extent can the mammalian visual system be shaped by visual behavior? Here we analyze the shape of the visual fields, the densities and distribution of cells in the retinal ganglion-cell layer and the organization of the visual projections in two species of facultative non-strictly subterranean rodents, Spalacopus cyanus and Ctenomys talarum, aiming to compare these traits with those of phylogenetically closely related species possessing contrasting diurnal/nocturnal visual habits. S. cyanus shows a definite zone of frontal binocular overlap and a corresponding area centralis, but a highly reduced amount of ipsilateral retinal projections. The situation in C. talarum is more extreme as it lacks of a fronto-ventral area of binocular superposition, has no recognizable area centralis and shows no ipsilateral retinal projections except to the suprachiasmatic nucleus. In both species, the extension of the monocular visual field and of the dorsal region of binocular overlap as well as the whole set of contralateral visual projections, appear well-developed. We conclude that these subterranean rodents exhibit, paradoxically, diurnal instead of nocturnal visual specializations, but at the same time suffer a specific regression of the anatomical substrate for stereopsis. We discuss these findings in light of the visual ecology of subterranean lifestyles.
Vega-Zuniga, T., Medina, F. S., Marín, G., Letelier, J. C., Palacios, A. G., Němec, P., Schleich, C. E., & Mpodozis, J. (2017)
Scientific Reports, 7, 41704. doi: 10.1038/srep41704.
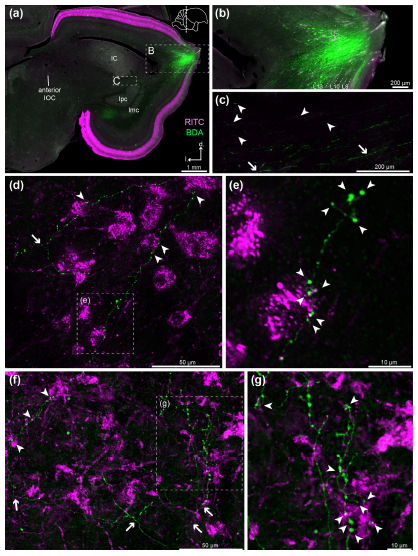
Abstract. The avian centrifugal visual system, which projects from the brain to the retina, has been intensively studied in several Neognathous birds that have a distinct isthmo‐optic nucleus (ION). However, birds of the order Palaeognathae seem to lack a proper ION in histologically stained brain sections. We had previously reported in the palaeognathous Chilean Tinamou (Nothoprocta perdicaria) that intraocular injections of Cholera Toxin B subunit retrogradely label a considerable number of neurons, which form a diffuse isthmo‐optic complex (IOC). In order to better understand how this IOC‐based centrifugal visual system is organized, we have studied its major components by means of in vivo and in vitro tracing experiments. Our results show that the IOC, though structurally less organized than an ION, possesses a dense core region consisting of multipolar neurons. It receives afferents from neurons in L10a of the optic tectum, which are distributed with a wider interneuronal spacing than in Neognathae. The tecto‐IOC terminals are delicate and divergent, unlike the prominent convergent tecto‐ION terminals in Neognathae. The centrifugal IOC terminals in the retina are exclusively divergent, resembling the terminals from “ectopic” centrifugal neurons in Neognathae. We conclude that the Tinamou’s IOC participates in a comparable general IOC‐retina‐TeO‐IOC circuitry as the neognathous ION. However, the connections between the components are structurally different and their divergent character suggests a lower spatial resolution. Our findings call for further comparative studies in a broad range of species for advancing our understanding of the evolution, plasticity and functional roles of the avian centrifugal visual system.
Krabichler, Q., Vega-Zuniga, T., Carrasco, D., Fernandez, M., Gutiérrez-Ibáñez, C., Marín, G., & Luksch, H. (2017)
Journal of Comparative Neurology, 525: 2514–2534. doi: 10.1002/cne.24195.
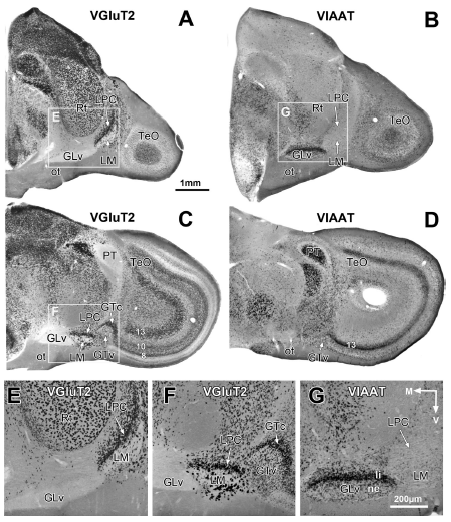
Abstract. The avian pretectal and ventrothalamic nuclei, encompassing the griseum tectale (GT), n. lentiformis mesencephali (LM), and n. geniculatus lateralis pars ventralis (GLv), are prominent retinorecipient structures related to optic flow operations and visuomotor control. Hence, a close coordination of these neural circuits is to be expected. Yet the connectivity among these nuclei is poorly known. Here, using intracellular labeling and in situ hybridization, we investigated the detailed morphology, connectivity, and neurochemical identity of neurons in these nuclei. Two different cell types exist in the GT: one that generates an axonal projection to the optic tectum (TeO), LM, GLv, and n. intercalatus thalami (ICT), and a second population that only projects to the LM and GLv. In situ hybridization revealed that most neurons in the GT express the vesicular glutamate transporter (VGluT2) mRNA, indicating a glutamatergic identity. In the LM, three morphological cell types were defined, two of which project axons towards dorsal targets. The LM neurons showed strong VGluT2 expression. Finally, the cells located in the GLv project to the TeO, LM, GT, n. principalis precommisuralis (PPC), and ICT. All neurons in the GLv showed strong expression of the vesicular inhibitory amino acid transporter (VIAAT) mRNA, suggesting a GABAergic identity. Our results show that the pretectal and ventrothalamic nuclei are highly interconnected, especially by glutamatergic and GABAergic neurons from the GT and GLv, respectively. This complex morphology and connectivity might be required to organize orienting visuomotor behaviors and coordinate the specific optic flow patterns that they induce.
Vega-Zuniga, Tomas, Marín, G., González-Cabrera, C., Planitscher, E., Hartmann, A., Marks, V., Mpodozis, J., & Luksch, H. (2016)
Journal of Comparative Neurology, 524: 2208-2229. doi: 10.1002/cne.23941.
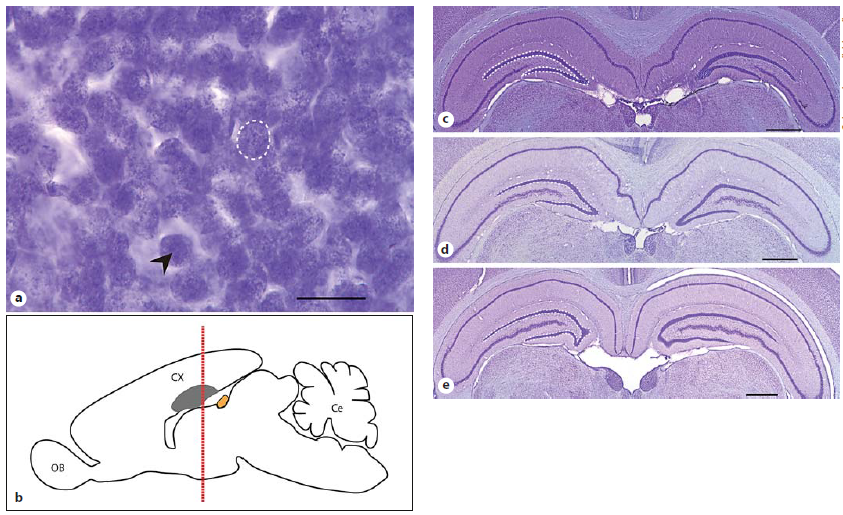
Abstract. Navigational and social challenges due to habitat conditions and sociality are known to influence dentate gyrus (DG) morphology, yet the relative importance of these factors remains unclear. Thus, we studied three natural populations of O. lunatus (Los Molles) and Octodon degus (El Salitre and Rinconada), two caviomorph species that differ in the extent of sociality and with contrasting vegetation cover of habitat used. The brains and DG of male and female breeding degus with simultaneous information on their physical and social environments were examined. The extent of sociality was quantified from total group size and range area overlap. O. degus at El Salitre was more social than at Rinconada and than O. lunatus from Los Molles. The use of transects to quantify cover of vegetation (and other physical objects in the habitat) and measures of the spatial behavior of animals indicated animal navigation based on unique cues or global landmarks is more cognitively challenging to O. lunatus. During lactation, female O. lunatus had larger brains than males. Relative DG volume was similar across sexes and populations. The right hemisphere of male and female O. lunatus had more cells than the left hemisphere, with DG directional asymmetry not found in O. degus. Degu population differences in brain size and DG cell number seemed more responsive to differences in habitat than to differences in sociality. Yet, large-sized O. degus (but not O. lunatus) that ranged over larger areas and were members of larger social groups had more DG cells per hemisphere. Thus, within-population variation in DG cell number by hemisphere was consistent with a joint influence of habitat and sociality in O. degus at El Salitre.
Sobrero, R., Fernández-Aburto, P., Ly-Prieto, Á., Delgado, S. E., Mpodozis, J., & Ebensperger, L. A. (2016)
Brain, Behavior and Evolution, 87: 51-64. doi: 10.1159/000444741.
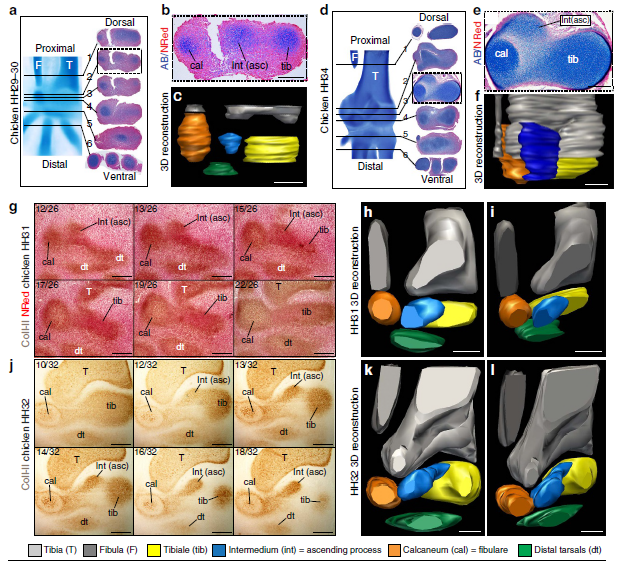
The anklebone (astragalus) of dinosaurs presents a characteristic upward projection, the ‘ascending process’ (ASC). The ASC is present in modern birds, but develops a separate ossification centre, and projects from the calcaneum in most species. These differences have been argued to make it non-comparable to dinosaurs. We studied ASC development in six different orders of birds using traditional techniques and spin–disc microscopy for whole-mount immunofluorescence. Unexpectedly, we found the ASC derives from the embryonic intermedium, an ancient element of the tetrapod ankle. In some birds it comes in contact with the astragalus, and, in others, with the calcaneum. The fact that the intermedium fails to fuse early with the tibiale and develops an ossification centre is unlike any other amniotes, yet resembles basal, amphibian-grade tetrapods. The ASC originated in early dinosaurs along changes to upright posture and locomotion, revealing an intriguing combination of functional innovation and reversion in its evolution.
Ossa-Fuentes, L., Mpodozis, J., & Vargas, A. O. (2015)
Nature Communications, 6, 8902: 1-7. doi: 10.1038/ncomms9902.
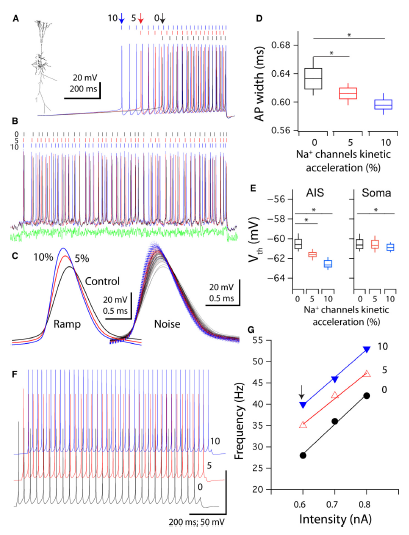
Abstract. Copper, an ion with many important metabolic functions, has also been proposed to have a role as modulator on neuronal function, mostly based on its effects on voltage‐ and neurotransmitter‐gated conductance as well as on neurological symptoms of patients with altered copper homeostasis. Nevertheless, the mechanisms by which copper exerts its neuromodulatory effects have not been clearly established in a functional neuronal network. Using rat hippocampus slices as a neuronal network model, the effects of copper in the range of 10–100 nm were tested on the intrinsic, synaptic and network properties of the CA1 region. Most of the previously described effects of this cation were in the micromolar range of copper concentrations. The current results indicate that copper is a multifaceted neuromodulator, having effects that may be grouped into two categories: (i) activity enhancement, by modulating synaptic communication and action potential (AP) conductances; and (ii) temporal processing and correlation extraction, by improving reliability and depressing inhibition. Specifically it was found that copper hyperpolarizes AP firing threshold, enhances neuronal and network excitability, modifies CA3–CA1 pathway gain, enhances the frequency of spontaneous synaptic events, decreases inhibitory network activity, and improves AP timing reliability. Moreover, copper chelation by bathocuproine decreases spontaneous network spiking activity. These results allow the proposal that copper affects the network activity from cellular to circuit levels on a moment‐by‐moment basis, and should be considered a crucial functional component of hippocampal neuronal circuitry.
Maureira, C., Letelier, J. C., Alvarez, O., Delgado, R., & Vergara, C. (2015)
European Journal of Neuroscience, 42: 3066-3080. doi: 10.1111/ejn.13104.
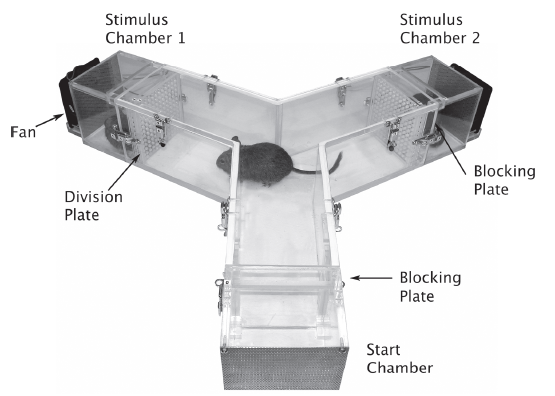
Abstract. We evaluated the extent to which manipulation of early olfactory environment can influence social behaviours in the South American Hystricognath rodent Octodon degus. The early olfactory environment of newborn degus was manipulated by scenting all litter members with eucalyptol during the first month of life. The social behaviour of sexually mature animals (5–7 months old) towards conspecifics was then assessed using a y-maze to compare the response of control (naïve) and treated animals to two different olfactory configurations (experiment 1): (i) a non-familiarized conspecific impregnated with eucalyptol (eucalyptol arm) presented against (ii) a non-familiarized unscented conspecific (control arm). In addition, in dyadic encounters, we assessed the behaviour of control and eucalyptol treated animals towards a non-familiarized conspecific scented with eucalyptol (experiment 2). We found that control subjects explored and spent significantly less time in the eucalyptol arm, indicating neophobic behaviours towards the artificially scented conspecific. Treated subjects explored and spent similar time in both arms of the maze, showing the same interest for both olfactory stimuli presented. During dyadic encounters in experiment 2, an interaction effect between early experience and sex was observed. Control males escaped and avoided their scented partner more frequently than eucalyptol treated male subjects and than females. Both groups did not differ in the exploration of their scented partners, suggesting that avoidance within agonistic context does not relate to neophobic behaviours. Our results suggest that the exposure to eucalyptol during early ontogeny decreases evasive behaviours within an agonistic context as a result of olfactory learning. Altogether, these results indicate that olfactory cues learned in early ontogeny can influence olfactory-guided behaviours in adult degus.
Márquez, N., Martínez-Harms, J., Vásquez, R. A., & Mpodozis, J. (2015)
PLoS ONE 10(2): e0118018, 1-12. doi: 10.1371/journal.pone.0118018.
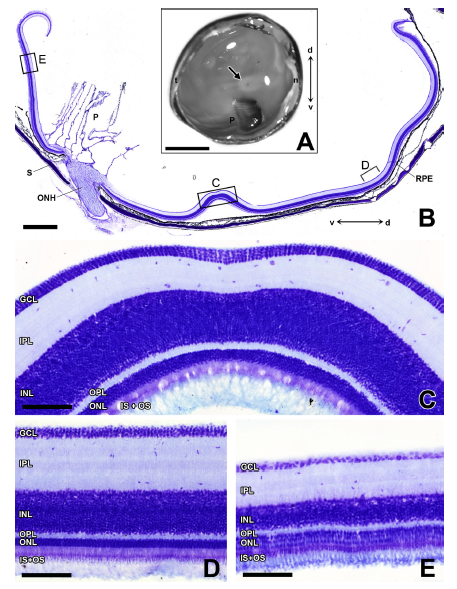
Abstract. Most systematic studies of the avian visual system have focused on Neognathous species, leaving virtually unexplored the Palaeognathae, comprised of the flightless ratites and the South American tinamous. We investigated the visual field, the retinal topography, and the pattern of retinal and centrifugal projections in the Chilean tinamou, a small Palaeognath of the family Tinamidae. The tinamou has a panoramic visual field with a small frontal binocular overlap of 20°. The retina possesses three distinct topographic specializations: a horizontal visual streak, a dorsotemporal area, and an area centralis with a shallow fovea. The maximum ganglion cell density is 61,900/ mm2, comparable to Falconiformes. This would provide a maximal visual acuity of 14.0 cycles/degree, in spite of relatively small eyes. The central retinal projections generally conform to the characteristic arrangement observed in Neognathae, with well‐differentiated contralateral targets and very few ipsilateral fibers. The centrifugal visual system is composed of a considerable number of multipolar centrifugal neurons, resembling the “ectopic” neurons described in Neognathae. They form a diffuse nuclear structure, which may correspond to the ancestral condition shared with other sauropsids. A notable feature is the presence of terminals in deep tectal layers 11–13. These fibers may represent either a novel retinotectal pathway or collateral branches from centrifugal neurons projecting to the retina. Both types of connections have been described in chicken embryos. Our results widen the basis for comparative studies of the vertebrate visual system, stressing the conserved character of the visual projections’ pattern within the avian clade.
Krabichler, Q., Vega-Zuniga, T., Morales, C., Luksch, H., & Marín, G. J. (2015)
Journal of Comparative Neurology, 523: 226-250. doi: 10.1002/cne.23676.
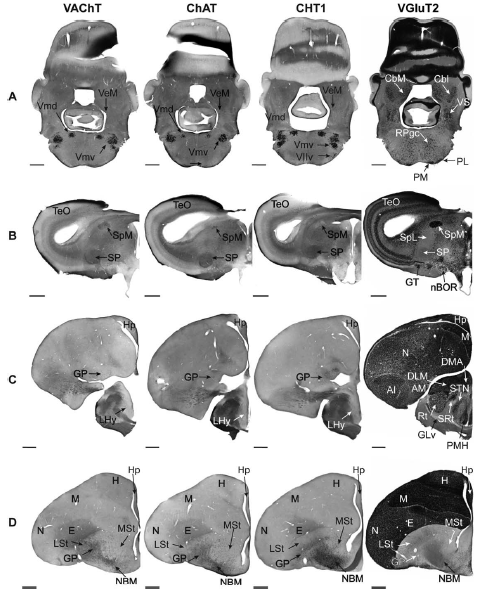
Abstract. Retinal inputs to the optic tectum (TeO) triggered by moving stimuli elicit synchronized feedback signals from two isthmic nuclei: the isthmi parvocelullaris (Ipc) and isthmi semilunaris (SLu). Both of these nuclei send columnar axon terminals back to the same tectal position receiving the retinal input. The feedback signals from the Ipc seem to act as an attentional spotlight by selectively boosting the propagation of retinal inputs from the tectum to higher visual areas. Although Ipc and SLu nuclei are widely considered cholinergic because of their immunoreactivity for choline acetyltransferase (ChAT), contradictory findings, including the expression of the vesicular glutamate transporter 2 (VGluT2) mRNA in Ipc neurons, have raised doubts about the purely cholinergic nature of this nucleus. In this study, in chicks, we revise the neurochemical identity of the isthmic nuclei by using in situ hybridization assays for VGluT2 along with three cholinergic markers: the vesicular acetylcholine transporter (VAChT), the high‐affinity choline transporter (CHT1) and ChAT. We found that neurons in the SLu showed strong mRNA expression of all three cholinergic markers, whereas the expression of VAChT mRNA in the Ipc was undetectable in our essays. Instead, Ipc neurons exhibited a strong expression of VGluT2 mRNA. Immunohistochemistry assays showed VGluT2 immunoreactivity in the TeO codistributing with anterogradely labeled Ipc axon‐terminal boutons, further supporting a glutamatergic function for the Ipc nucleus. Therefore, our results strongly suggest that, in the chick, whereas the feedback from the SLu to the TeO is indeed cholinergic, the feedback from the Ipc has a marked glutamatergic component.
González-Cabrera, C., Garrido-Charad, F., Roth, A., & Marín, G. J. (2015)
Journal of Comparative Neurology, 523: 1341-1358. doi: 10.1002/cne.23739.
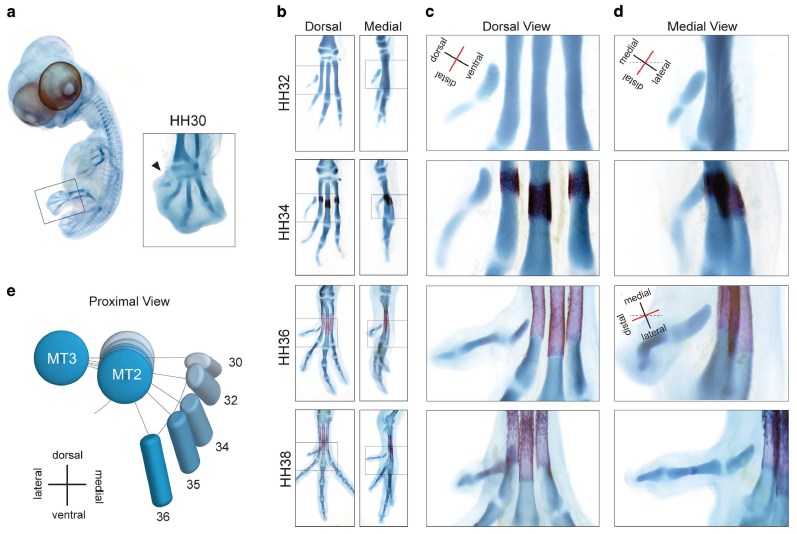
Abstract. Most birds have an opposable digit 1 (hallux) allowing the foot to grasp, which evolved from the non-opposable hallux of early theropod dinosaurs. An important morphological difference with early theropods is the twisting of the long axis of its metatarsal. Here, we show how embryonic musculature and the onset of its activity are required for twisting of metatarsal 1 (Mt1) and retroversion of the hallux. Pharmacologically paralyzed embryos do not fully retrovert the hallux and have a straight Mt1 shaft, phenocopying the morphology of early tetanuran dinosaurs. Molecular markers of cartilage maturation and ossification show that differentiation of Mt1 is significantly delayed compared to Mt2-4. We hypothesize on how delayed maturation may have increased plasticity, facilitating muscular twisting. Our experimental results emphasize the importance of embryonic muscular activity in the evolutionary origin of a crucial adaptation.
Francisco Botelho, J., Smith-Paredes, D., Soto-Acuña, S., Mpodozis, J., Palma, V., & Vargas, A. O. (2015)
Scientific Reports, 5, 9840: 1-11. doi: 10.1038/srep09840.
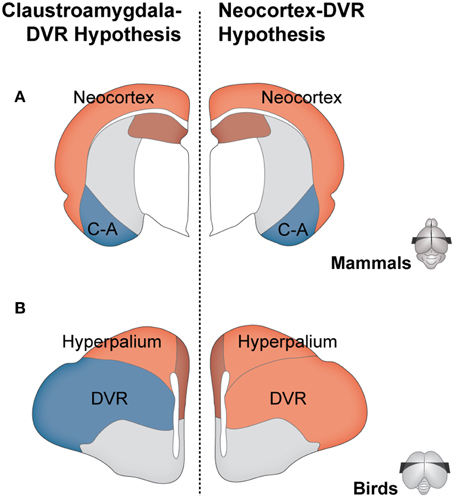
Abstract. Owen’s pre-evolutionary definition of a homolog as “the same organ in different animals under every variety of form and function” and its redefinition after Darwin as “the same trait in different lineages due to common ancestry” entail the same heuristic problem: how to establish “sameness.”Although different criteria for homology often conflict, there is currently a generalized acceptance of gene expression as the best criterion. This gene-centered view of homology results from a reductionist and preformationist concept of living beings. Here, we adopt an alternative organismic-epigenetic viewpoint, and conceive living beings as systems whose identity is given by the dynamic interactions between their components at their multiple levels of composition. We posit that there cannot be an absolute homology criterion, and instead, homology should be inferred from comparisons at the levels and developmental stages where the delimitation of the compared trait lies. In this line, we argue that neural connectivity, i.e., the hodological criterion, should prevail in the determination of homologies between brain supra-cellular structures, such as the vertebrate pallium.
Faunes, M., Botelho, J. F., Galleguillos, P. A., & Mpodozis, J. (2015)
Frontiers in Neuroscience, 9, 223, 1-7. doi: 10.3389/fnins.2015.00223.
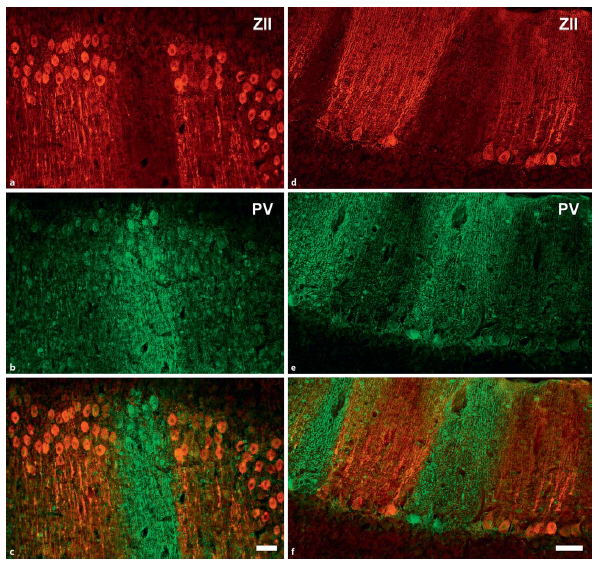
Abstract. Zebrin II (ZII) is a glycolytic enzyme expressed in cerebellar Purkinje cells. In both mammals and birds, ZII is expressed heterogeneously, such that there are sagittal stripes of Purkinje cells with a high ZII expression (ZII+) alternating with stripes of Purkinje cells with little or no expression (ZII-). To date, ZII expression studies are limited to neognathous birds: pigeons (Columbiformes), chickens (Galliformes), and hummingbirds (Trochilidae). These previous studies divided the avian cerebellum into 5 transverse regions based on the pattern of ZII expression. In the lingular region (lobule I) all Purkinje cells are ZII+. In the anterior region (lobules II-V) there are 4 pairs of ZII+/- stripes. In the central region (lobules VI-VIII) all Purkinje cells are ZII+. In the posterior region (lobules VIII-IX) there are 5-7 pairs of ZII+/- stripes. Finally, in the nodular region (lobule X) all Purkinje cells are ZII+. As the pattern of ZII stripes is quite similar in these disparate species, it appears that it is highly conserved. However, it has yet to be studied in paleognathous birds, which split from the neognaths over 100 million years ago. To better understand the evolution of cerebellar compartmentation in birds, we examined ZII immunoreactivity in a paleognath, the Chilean tinamou (Nothoprocta perdicaria). In the tinamou, Purkinje cells expressed ZII heterogeneously such that there were sagittal ZII+ and ZII- stripes of Purkinje cells, and this pattern of expression was largely similar to that observed in neognathous birds. For example, all Purkinje cells in the lingular (lobule I) and nodular (lobule X) regions were ZII+, and there were 4 pairs of ZII+/- stripes in the anterior region (lobules II-V). In contrast to neognaths, however, ZII was expressed in lobules VI-VII as a series of sagittal stripes in the tinamou. Also unlike in neognaths, stripes were absent in lobule IXab, and all Purkinje cells expressed ZII in the tinamou. The differences in ZII expression between the tinamou and neognaths could reflect behavior, but the general similarity of the expression patterns across all bird species suggests that ZII stripes evolved early in the avian phylogenetic tree.
Corfield, J. R., Kolominsky, J., Marin, G. J., Craciun, I., Mulvany-Robbins, B. E., Iwaniuk, A. N., & Wylie, D. R. (2015)
Brain, Behavior and Evolution, 85: 94-106. doi: 10.1159/000380810.
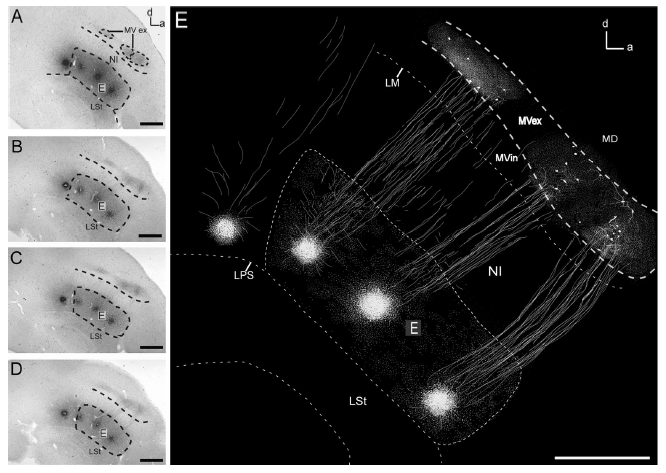
Abstract. The dorsal ventricular ridge (DVR) is one of the main components of the sauropsid pallium. In birds, the DVR is formed by an inner region, the nidopallium, and a more dorsal region, the mesopallium. The nidopallium contains discrete areas that receive auditory, visual, and multisensory collothalamic projections. These nidopallial nuclei are known to sustain reciprocal, short‐range projections with their overlying mesopallial areas. Recent findings on the anatomical organization of the auditory DVR have shown that these short‐range projections have a columnar organization that closely resembles that of the mammalian neocortex. However, it is unclear whether this columnar organization generalizes to other areas within the DVR. Here we examine in detail the organization of the visual DVR, performing small, circumscribed deposits of neuronal tracers as well as intracellular fillings in brain slices. We show that the visual DVR is organized in three main laminae, the thalamorecipient nucleus entopallium; a dorsally adjacent nidopallial lamina, the intermediate nidopallium; and a contiguous portion of the ventral mesopallium, the mesopallium ventrale. As in the case of the auditory DVR, we found a highly topographically organized system of reciprocal interconnections among these layers, which was formed by dorsoventrally oriented, discrete columnar bundles of axons. We conclude that the columnar organization previously demonstrated in the auditory DVR is not a unique feature but a general characteristic of the avian sensory pallium. We discuss these results in the context of a comparison between sauropsid and mammalian pallial organization.
Ahumada-Galleguillos, P., Fernández, M., Marin, G. J., Letelier, J. C., & Mpodozis, J. (2015)
Journal of Comparative Neurology, 523: 2618-2636. doi: 10.1002/cne.23808.
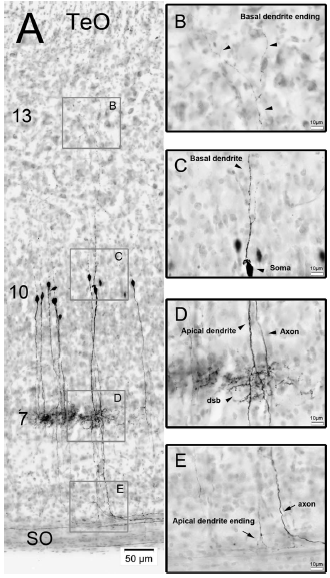
Abstract. The nucleus geniculatus lateralis pars ventralis (GLv) is a prominent retinal target in all amniotes. In birds, it is in receipt of a dense and topographically organized retinal projection. The GLv is also the target of substantial and topographically organized projections from the optic tectum and the visual wulst (hyperpallium). Tectal and retinal afferents terminate homotopically within the external GLv‐neuropil. Efferents from the GLv follow a descending course through the tegmentum and can be traced into the medial pontine nucleus. At present, the cells of origin of the Tecto‐GLv projection are only partially described. Here we characterized the laminar location, morphology, projection pattern, and neurochemical identity of these cells by means of neural tracer injections and intracellular fillings in slice preparations and extracellular tracer injections in vivo. The Tecto‐GLv projection arises from a distinct subset of layer 10 bipolar neurons, whose apical dendrites show a complex transverse arborization at the level of layer 7. Axons of these bipolar cells arise from the apical dendrites and follow a course through the optic tract to finally form very fine and restricted terminal endings inside the GLv‐neuropil. Double‐label experiments showed that these bipolar cells were choline acetyltransferase (ChAT)‐immunoreactive. Our results strongly suggest that Tecto‐GLv neurons form a pathway by which integrated tectal activity rapidly feeds back to the GLv and exerts a focal cholinergic modulation of incoming retinal inputs.
Vega-Zuniga, Tomas, Mpodozis, J., Karten, H. J., Marín, G., Hain, S., & Luksch, H. (2014)
Journal of Comparative Neurology, 522: 2377-2396. doi:10.1002/cne.23539.
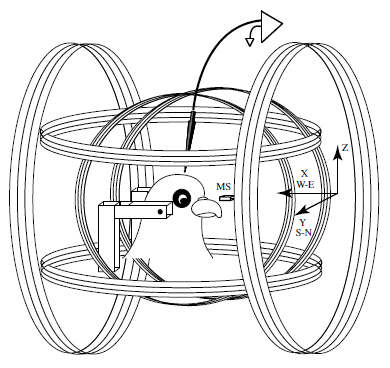
Abstract. There is a consensus that birds detect the earth’s magnetic field and use some of its features for orientation and homing purposes. Since the late 1960s, when the first solid behavioral evidence of magnetoreception was obtained, much research has been devoted to describing the ethological aspects of this behavior. The neurophysiological basis of magnetoreception has been much less studied, although a frequently cited 1986 report described a high prevalence (70 %) of magneto-sensitive neurons in the pigeon optic tectum with high signal-to-noise ratios (Semm and Demaine, J Comp Physiol A 159:619–625, 1986). Here, we repeated these neurophysiological experiments using anesthetized as well as awake pigeons and new recording techniques. Our data indicate that magneto-sensitive units do not exist in the avian tectum.
Ramírez, E., Marín, G., Mpodozis, J., & Letelier, J. C. (2014)
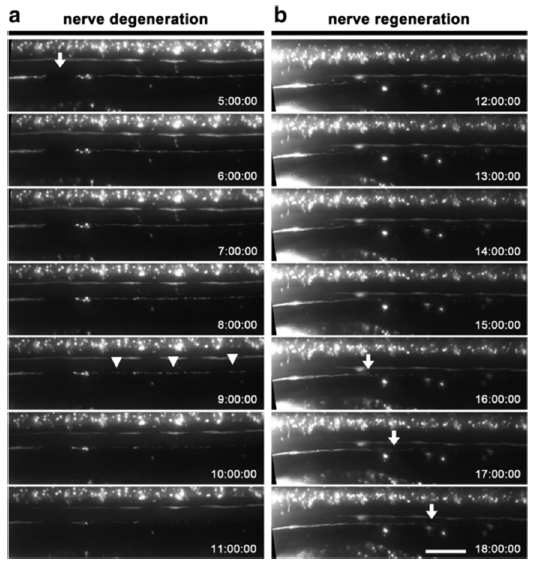
Abstract. Background: Tissue injury has been employed to study diverse biological processes such as regeneration and inflammation. In addition to physical or surgical based methods for tissue injury, current protocols for localized tissue damage include laser and two-photon wounding, which allow a high degree of accuracy, but are expensive and difficult to apply. In contrast, electrical injury is a simple and inexpensive technique, which allows reproducible and localized cell or tissue damage in a variety of contexts. Results: We describe a novel technique that combines the advantages of zebrafish for in vivo visualization of cells with those of electrical injury methods in a simple and versatile protocol which allows the study of regeneration and inflammation. The source of the electrical pulse is a microelectrode that can be placed with precision adjacent to specific cells expressing fluorescent proteins. We demonstrate the use of this technique in zebrafish larvae by damaging different cell types and structures. Neurectomy can be carried out in peripheral nerves or in the spinal cord allowing the study of degeneration and regeneration of nerve fibers. We also apply this method for the ablation of single lateral line mechanosensory neuromasts, showing the utility of this approach as a tool for the study of organ regeneration. In addition, we show that electrical injury induces immune cell recruitment to damaged tissues, allowing in vivo studies of leukocyte dynamics during inflammation within a confined and localized injury. Finally, we show that it is possible to apply electroablation as a method of tissue injury and inflammation induction in adult fish. Conclusions: Electrical injury using a fine microelectrode can be used for axotomy of neurons, as a general tissue ablation tool and as a method to induce a powerful inflammatory response. We demonstrate its utility to studies in both larvae and in adult zebrafish but we expect that this technique can be readily applied to other organisms as well. We have called this method of electrical based tissue ablation, electroablation.
Moya-Díaz, J., Peña, O. A., Sánchez, M., Ureta, D. A., Reynaert, N. G., Anguita-Salinas, C., Marín, G., & Allende, M. L. (2014)
BMC Developmental Biology, 14, 7: 1-13. doi: 10.1186/1471-213X-14-7.
Abstract. In scatter-hoarding species, several behavioral and neuroanatomical adaptations allow them to store and retrieve thousands of food items per year. Nectarivorous animals face a similar scenario having to remember quality, location and replenishment schedules of several nectar sources. In the green-backed firecrown hummingbird (Sephanoides sephanoides), males are territorial and have the ability to accurately keep track of nectar characteristics of their defended food sources. In contrast, females display an opportunistic strategy, performing rapid intrusions into males territories. In response, males behave aggressively during the non-reproductive season. In addition, females have higher energetic demands due to higher thermoregulatory costs and travel times. The natural scenario of this species led us to compared cognitive abilities and hippocampal size between males and females. Males were able to remember nectar location and renewal rates significantly better than females. However, the hippocampal formation was significantly larger in females than males. We discuss these findings in terms of sexually dimorphic use of spatial resources and variable patterns of brain dimorphisms in birds.
González-Gómez, P. L., Madrid-Lopez, N., Salazar, J. E., Suárez, R., Razeto-Barry, P., Mpodozis, J., Bozinovic, F., & Vásquez, R. A. (2014)
PLoS ONE 9(3): e90165, 1-8. doi: 10.1371/journal.pone.0090165.
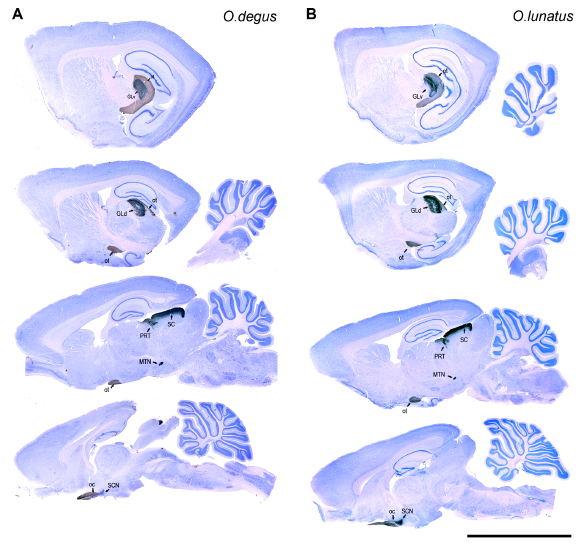
Abstract. Binocular vision is a visual property that allows fine discrimination of in-depth distance (stereopsis), as well as enhanced light and contrast sensitivity. In mammals enhanced binocular vision is structurally associated with a large degree of frontal binocular overlap, the presence of a corresponding retinal specialization containing a fovea or an area centralis, and well-developed ipsilateral retinal projections to the lateral thalamus (GLd). We compared these visual traits in two visually active species of the genus Octodon that exhibit contrasting visual habits: the diurnal Octodon degus, and the nocturnal Octodon lunatus. The O. lunatus visual field has a prominent 100° frontal binocular overlap, much larger than the 50° of overlap found in O. degus. Cells in the retinal ganglion cell layer were 40% fewer in O. lunatus (180,000) than in O. degus (300,000). O. lunatus has a poorly developed visual streak, but a well developed area centralis, located centrally near the optic disk (peak density of 4,352 cells/mm2). O. degus has a highly developed visual streak, and an area centralis located more temporally (peak density of 6,384 cells/mm2). The volumes of the contralateral GLd and superior colliculus (SC) are 15% larger in O. degus compared to O. lunatus. However, the ipsilateral projections to GLd and SC are 500% larger in O. lunatus than in O. degus. Other retinorecipient structures related to ocular movements and circadian activity showed no statistical differences between species. Our findings strongly suggest that nocturnal visual behavior leads to an enhancement of the structures associated with binocular vision, at least in the case of these rodents. Expansion of the binocular visual field in nocturnal species may have a beneficial effect in light and contrast sensitivity, but not necessarily in stereopsis. We discuss whether these conclusions can be extended to other mammalian and non-mammalian amniotes.
Vega-Zuniga, Tomas, Medina, F. S., Fredes, F., Zuniga, C., Severín, D., Palacios, A. G., Karten, H. J., & Mpodozis, J. (2013)
PLoS ONE 8(12): e84199, 1-12. doi: 10.1371/journal.pone.0084199.
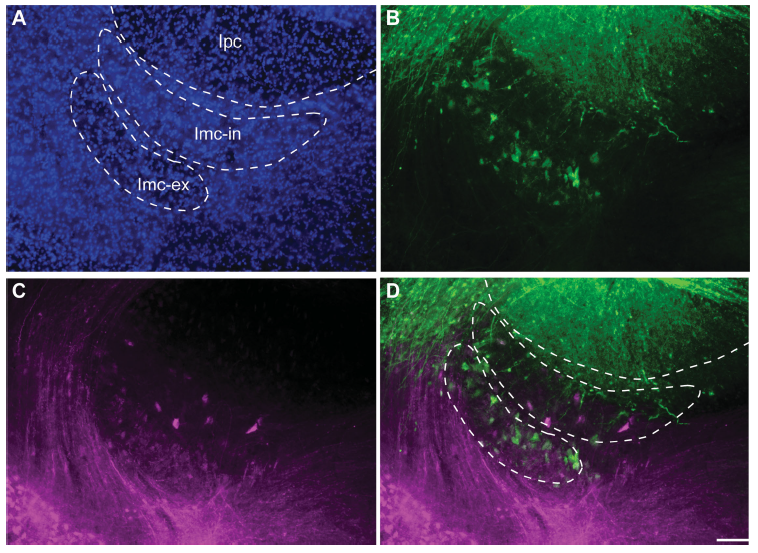
Abstract. The isthmic complex is part of a visual midbrain circuit thought to be involved in stimulus selection and spatial attention. In birds, this circuit is composed of the nuclei isthmi pars magnocellularis (Imc), pars parvocellularis (Ipc), and pars semilunaris (SLu), all of them reciprocally connected to the ipsilateral optic tectum (TeO). The Imc conveys heterotopic inhibition to the TeO, Ipc, and SLu via widespread γ‐aminobutyric acid (GABA)ergic axons that allow global competitive interactions among simultaneous sensory inputs. Anatomical studies in the chick have described a cytoarchitectonically uniform Imc nucleus containing two intermingled cell types: one projecting to the Ipc and SLu and the other to the TeO. Here we report that in passerine species, the Imc is segregated into an internal division displaying larger, sparsely distributed cells, and an external division displaying smaller, more densely packed cells. In vivo and in vitro injections of neural tracers in the TeO and the Ipc of the zebra finch demonstrated that neurons from the external and internal subdivisions project to the Ipc and the TeO, respectively, indicating that each Imc subdivision contains one of the two cell types hodologically defined in the chick. In an extensive survey across avian orders, we found that, in addition to passerines, only species of Piciformes and Rallidae exhibited a segregated Imc, whereas all other groups exhibited a uniform Imc. These results offer a comparative basis to investigate the functional role played by each Imc neural type in the competitive interactions mediated by this nucleus.
Faunes, M., Fernández, S., Gutiérrez-Ibáñez, C., Iwaniuk, A. N., Wylie, D. R., Mpodozis, J., Karten, H. J., & Marín, G. (2013)
Journal of Comparative Neurology, 521: 1727-1742. doi: 10.1002/cne.23253.
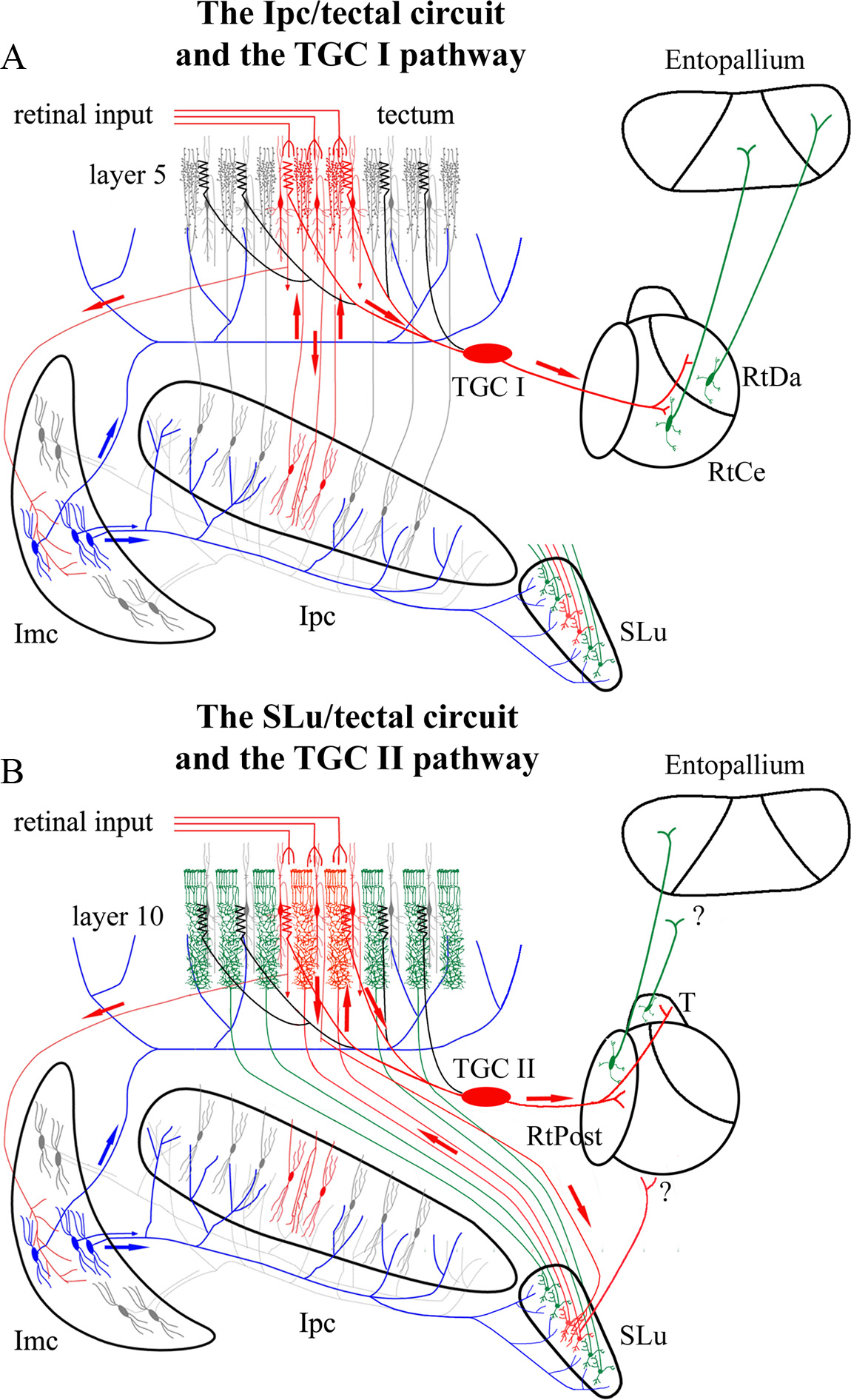
Abstract. When a salient object in the visual field captures attention, the neural representation of that object is enhanced at the expense of competing stimuli. How neural activity evoked by a salient stimulus evolves to take precedence over the neural activity evoked by other stimuli is a matter of intensive investigation. Here, we describe in pigeons (Columba livia) how retinal inputs to the optic tectum (TeO, superior colliculus in mammals), triggered by moving stimuli, are selectively relayed on to the rotundus (Rt, caudal pulvinar) in the thalamus, and to its pallial target, the entopallium (E, extrastriate cortex). We show that two satellite nuclei of the TeO, the nucleus isthmi parvocelullaris (Ipc) and isthmi semilunaris (SLu), send synchronized feedback signals across tectal layers. Preventing the feedback from Ipc but not from SLu to a tectal location suppresses visual responses to moving stimuli from the corresponding region of visual space in all Rt subdivisions. In addition, the bursting feedback from the Ipc imprints a bursting rhythm on the visual signals, such that the visual responses of the Rt and the E acquire a bursting modulation significantly synchronized to the feedback from Ipc. As the Ipc feedback signals are selected by competitive interactions, the visual responses within the receptive fields in the Rt tend to synchronize with the tectal location receiving the “winning” feedback from Ipc. We propose that this selective transmission of afferent activity combined with the cross-regional synchronization of the areas involved represents a bottom-up mechanism by which salient stimuli capture attention.
Marín, G. J., Durán, E., Morales, C., González-Cabrera, C., Sentis, E., Mpodozis, J., & Letelier, J. C. (2012)
Journal of Neuroscience, 32 (3), 1110-1122. doi: 10.1523/JNEUROSCI.4151-11.2012.
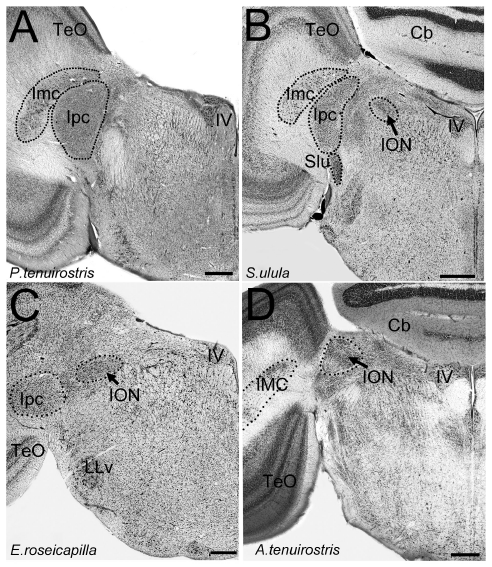
Abstract. In birds, there is a retinofugal projection from the brain to the retina originating from the isthmo optic nucleus (ION) in the midbrain. Despite a large number of anatomical, physiological and histochemical studies, the function of this retinofugal system remains unclear. Several functions have been proposed including: gaze stabilization, pecking behavior, dark adaptation, shifting attention, and detection of aerial predators. This nucleus varies in size and organization among some species, but the relative size and morphology of the ION has not been systematically studied. Here, we present a comparison of the relative size and morphology of the ION in 81 species of birds, representing 17 different orders. Our results show that several orders of birds, besides those previously reported, have a large, well-organized ION, including: hummingbirds, woodpeckers, coots and allies, and kingfishers. At the other end of the spectrum, parrots, herons, waterfowl, owls and diurnal raptors have relatively small ION volumes. ION also appears to be absent or unrecognizable is several taxa, including one of the basal avian groups, the tinamous, which suggests that the ION may have evolved only in the more modern group of birds, Neognathae. Finally, we demonstrate that evolutionary changes in the relative size and the cytoarchitectonic organization of ION have occurred largely independent of phylogeny. The large relative size of the ION in orders with very different lifestyles and feeding behaviors suggest there is no clear association with pecking behavior or predator detection. Instead, our results suggest that the ION is more complex and enlarged in birds that have eyes that are emmetropic in some parts of the visual field and myopic in others. We therefore posit that the ION is involved in switching attention between two parts of the retina i.e. from an emmetropic to a myopic part of the retina.
Gutiérrez-Ibáñez, C., Iwaniuk, A. N., Lisney, T. J., Faunes, M., Marín, G. J., & Wylie, D. R. (2012)
PLoS ONE 7(5): e37816, 1-14. doi: 10.1371/journal.pone.0037816.
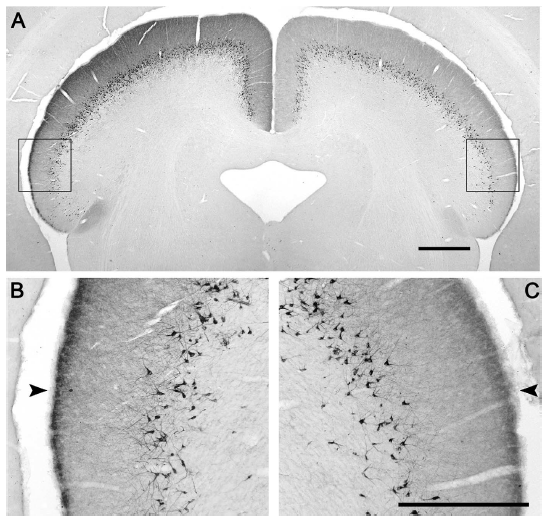
Abstract. The mammalian pulvinar complex is a collection of dorsal thalamic nuclei related to several visual and integrative processes. Previous studies have shown that the superficial layers of the superior colliculus project to multiple divisions of the pulvinar complex. Although most of these works agree about the existence of an ipsilateral tectopulvinar projection arising from the stratum griseum superficialis, some others report a bilateral projection originating from this same tectal layer. We investigated the organization of the tectopulvinar projections in the Californian ground squirrel using cholera toxin B (CTb). We confirmed previous studies showing that the caudal pulvinar of the squirrel receives a massive bilateral projection originating from a specific cell population located in the superficial collicular layers (SGS3, also called the “lower SGS” or “SGSL”). We found that this projection shares striking structural similarities with the tectorotundal pathway of birds and reptiles. Morphology of the collicular cells originating this projection closely corresponds to that of the bottlebrush tectal cells described previously for chickens and squirrels. In addition, we found that the rostral pulvinar receives an exclusively ipsilateral projection from a spatially separate population of collicular cells located at the base of the stratum opticum, deeper than the cells projecting to the caudal pulvinar. These results strongly support, at a structural level, the homology of the pathway originating in the SGS3 collicular cells upon the caudal pulvinar with the tectorotundal pathway of nonmammalian amniotes and contribute to clarifying the general organization of the tectopulvinar pathways in mammals.
Fredes, F., Vega-Zuniga, T., Karten, H., & Mpodozis, J. (2012)
Journal of Comparative Neurology, 520: 1800-1818. doi: 10.1002/cne.23014.
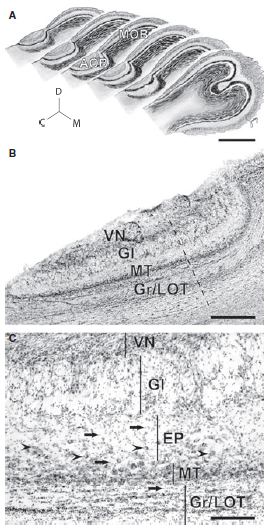
Abstract. The vomeronasal system is crucial for social and sexual communication in mammals. Two populations of vomeronasal sensory neurons, each expressing Gαi2 or Gαo proteins, send projections to glomeruli of the rostral or caudal accessory olfactory bulb, rAOB and cAOB, respectively. In rodents, the Gαi2‐ and Gαo‐expressing vomeronasal pathways have shown differential responses to small/volatile vs. large/non‐volatile semiochemicals, respectively. Moreover, early gene expression suggests predominant activation of rAOB and cAOB neurons in sexual vs. aggressive contexts, respectively. We recently described the AOB of Octodon degus, a semiarid‐inhabiting diurnal caviomorph. Their AOB has a cell indentation between subdomains and the rAOB is twice the size of the cAOB. Moreover, their AOB receives innervation from the lateral aspect, contrasting with the medial innervation of all other mammals examined to date. Aiming to relate AOB anatomy with lifestyle, we performed a morphometric study on the AOB of the capybara, a semiaquatic caviomorph whose lifestyle differs remarkably from that of O. degus. Capybaras mate in water and scent‐mark their surroundings with oily deposits, mostly for male–male communication. We found that, similar to O. degus, the AOB of capybaras shows a lateral innervation of the vomeronasal nerve, a cell indentation between subdomains and heterogeneous subdomains, but in contrast to O. degus the caudal portion is larger than the rostral one. We also observed that four other caviomorph species present a lateral AOB innervation and a cell indentation between AOB subdomains, suggesting that those traits could represent apomorphies of the group. We propose that although some AOB traits may be phylogenetically conserved in caviomorphs, ecological specializations may play an important role in shaping the AOB.
Suárez, R., Santibáñez, R., Parra, D., Coppi, A. A., Abrahão, L. M. B., Sasahara, T. H. C., & Mpodozis, J. (2011)
Journal of Anatomy, 218: 558-565. doi: 10.1111/j.1469-7580.2011.01357.x.
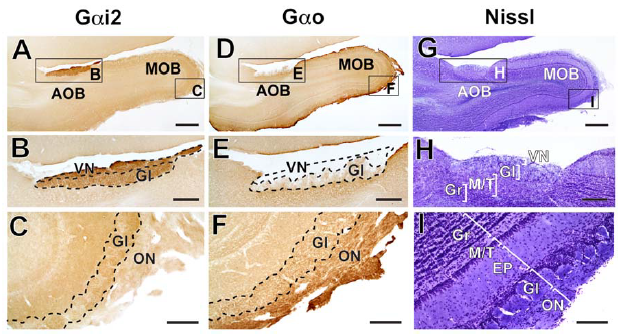
Abstract. In mammals, social and sexual behaviours are largely mediated by the vomeronasal system (VNS). The accessory olfactory bulb (AOB) is the first synaptic locus of the VNS and ranges from very large in Caviomorph rodents, small in carnivores and ungulates, to its complete absence in apes, elephants, most bats and aquatic species. Two pathways have been described in the VNS of mammals. In mice, vomeronasal neurons expressing Gαi2 protein project to the rostral portion of the AOB and respond mostly to small volatile molecules, whereas neurons expressing Gαo project to the caudal AOB and respond mostly to large non-volatile molecules. However, the Gαo-expressing pathway is absent in several species (horses, dogs, musk shrews, goats and marmosets) but no hypotheses have been proposed to date to explain the loss of that pathway. We noted that the species that lost the Gαo pathway belong to Laurasiatheria and Primates lineages, both clades with ubiquitous sexual dimorphisms across species. To assess whether similar events of Gαo pathway loss could have occurred convergently in dimorphic species we studied G-protein expression in the AOB of two species that independently evolved sexually dimorphic traits: the California ground squirrel Spermophilus beecheyi (Rodentia; Sciurognathi) and the cape hyrax Procavia capensis (Afrotheria; Hyracoidea). We found that both species show uniform expression of Gαi2-protein throughout AOB glomeruli, while Gαo expression is restricted to main olfactory glomeruli only. Our results suggest that the degeneration of the Gαo-expressing vomeronasal pathway has occurred independently at least four times in Eutheria, possibly related to the emergence of sexual dimorphisms and the ability of detecting the gender of conspecifics at distance.
Suárez, R., Fernández-Aburto, P., Manger, P. R., & Mpodozis, J. (2011)
PLoS ONE 6(10): e26436, 1-6. doi: 10.1371/journal.pone.0026436
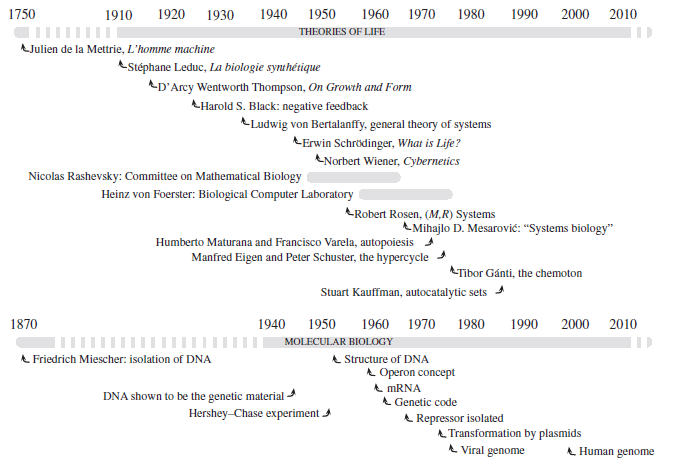
Abstract. The nature of life has been a topic of interest from the earliest of times, and efforts to explain it in mechanistic terms date at least from the 18th century. However, the impressive development of molecular biology since the 1950s has tended to have the question put on one side while biologists explore mechanisms in greater and greater detail, with the result that studies of life as such have been confined to a rather small group of researchers who have ignored one another’s work almost completely, often using quite different terminology to present very similar ideas. Central among these ideas is that of closure, which implies that all of the catalysts needed for an organism to stay alive must be produced by the organism itself, relying on nothing apart from food (and hence chemical energy) from outside. The theories that embody this idea to a greater or less degree are known by a variety of names, including (M,R) systems, autopoiesis, the chemoton, the hypercycle, symbiosis, autocatalytic sets, sysers and RAF sets. These are not all the same, but they are not completely different either, and in this review we examine their similarities and differences, with the aim of working towards the formulation of a unified theory of life.
Letelier, J. C., Cárdenas, M. L., & Cornish-Bowden, A. (2011)
Journal of Theoretical Biology, 286: 100-113. doi: 10.1016/j.jtbi.2011.06.033.
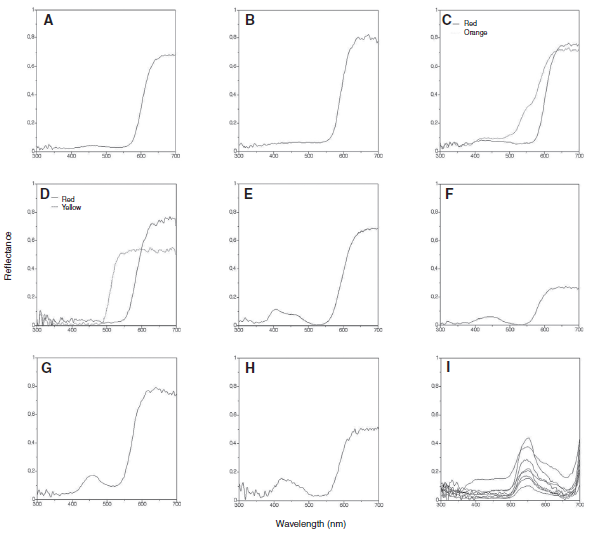
Abstract. It has been argued that trichromatic bees with photoreceptor spectral sensitivity peaks in the ultraviolet (UV), blue and green areas of the spectrum are blind to long wavelengths (red to humans). South American temperate forests (SATF) contain a large number of human red-looking flowers that are reported to be visited by the bumblebee Bombus dahlbomii. In the present study, B. dahlbomii’s spectral sensitivity was measured through electroretinogram (ERG) recordings. No extended sensitivity to long wavelengths was found in B. dahlbomii. The spectral reflectance curves from eight plant species with red flowers were measured. The color loci occupied by these flowers in the bee color space was evaluated using the receptor noise-limited model. Four of the plant species have pure red flowers with low levels of chromatic contrast but high levels of negative L-receptor contrast. Finally, training experiments were performed in order to assess the role of achromatic cues in the detection and discrimination of red targets by B. dahlbomii. The results of the training experiments suggest that the bumblebee relies on achromatic contrast provided by the L-receptor to detect and discriminate red targets. These findings are discussed in the context of the evolutionary background under which the relationship between SATF species and their flower visitors may have evolved.
Martínez-Harms, J., Palacios, A. G., Márquez, N., Estay, P., Arroyo, M. T. K., & Mpodozis, J. (2010)
Journal of Experimental Biology, 213: 564-571. doi: 10.1242/jeb.037622.
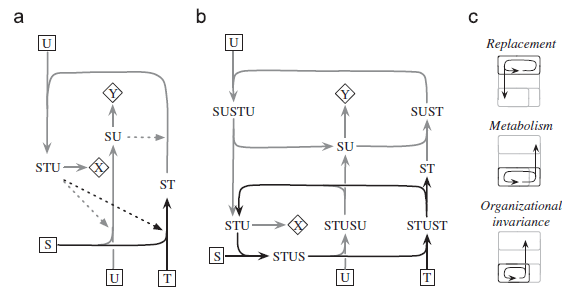
Abstract. The major insight in Robert Rosen’s view of a living organism as an (M,R)-system was the realization that an organism must be “closed to efficient causation”, which means that the catalysts needed for its operation must be generated internally. This aspect is not controversial, but there has been confusion and misunderstanding about the logic Rosen used to achieve this closure. In addition, his corollary that an organism is not a mechanism and cannot have simulable models has led to much argument, most of it mathematical in nature and difficult to appreciate. Here we examine some of the mathematical arguments and clarify the conditions for closure.
Luz Cárdenas, M., Letelier, J. C., Gutierrez, C., Cornish-Bowden, A., & Soto-Andrade, J. (2010)
Journal of Theoretical Biology, 263, 1: 79-92. doi: 10.1016/j.jtbi.2009.11.010.
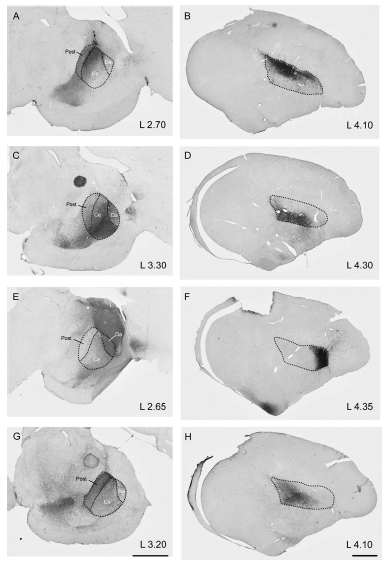
Abstract. The tectofugal pathway (retina – optic tectum – nucleus rotundus – entopallium) is a prominent route mediating visual discrimination in diurnal birds. Several lines of evidence have shown that at the tecto‐rotundal stage this pathway is composed of multiple parallel channels. Anatomical studies show that the nucleus rotundus is composed of at least four subdivisions, according to differences in cytoarchitectonic, histochemical, and hodological properties. Each of these subdivisions is in receipt of a highly convergent, nontopographic tectal projection, originating from a distinct subset of tecto‐rotundal neurons. Physiological studies show that neurons of different subdivisions respond specifically to different visual dimensions, such as color, luminance, two‐dimensional motion, and in‐depth motion. At present it is less clear whether or to what extent this channel segregation is preserved at the telencephalic stage of the tectofugal pathway. The entopallium shows no obvious subdivisions or laminations. Nevertheless, tract‐tracing experiments show that separate portions of the entopallium receive efferent projections from different rotundal subdivisions, in a way that maintains the rostrocaudal order of these subdivisions. In the present study we investigate in detail the topography of the rotundo‐entopallial projection by means of anterograde and retrograde neuronal tracers. Our results confirm the zonal topography proposed by previous studies and indicate that each zone in the entopallium receives a direct and topographically organized projection from its corresponding rotundal subdivision. These results suggest that the spatial arrangement of the different rotundal functional modules is preserved at the entopallial level.
Fredes, F., Tapia, S., Letelier, J. C., Marín, G., & Mpodozis, J. (2010)
Journal of Comparative Neurology, 518: 4342-4361. doi: 10.1002/cne.22460.
Lista Completa
A specialized reciprocal connectivity suggests a link between the mechanisms by which the superior colliculus and parabigeminal nucleus produce defensive behaviors in rodents.
Deichler, A., Carrasco, D., Lopez-Jury, L., Vega-Zuniga, T., Márquez, N., Mpodozis, J., & Marín, G. J. (2020)
Scientific Reports, 10, 16220: 1-16. doi: 10.1038/s41598-020-72848-0.
The evolutionary consequences of epigenesis and neutral change: A conceptual approach at the organismal level.
Vargas, A. O., Botelho, J. F. & Mpodozis, J. (2020)
The Journal of Experimental Zoology Part B: Molecular and Developmental Evolution: 1– 10. doi: 10.1002/jez.b.23023.
Parallel organization of the avian sensorimotor arcopallium: Tectofugal visual pathway in the pigeon (Columba livia).
Fernández, M., Morales, C., Durán, E., Fernández-Colleman, S., Sentis, E., Mpodozis, J., Karten, H. J., & Marín, G. J. (2020)
Journal of Comparative Neurology, 528: 597-623. doi: 10.1002/cne.24775.
Intratelencephalic projections of the avian visual dorsal ventricular ridge: Laminarly segregated, reciprocally and topographically organized.
Fernández, M., Ahumada-Galleguillos, P., Sentis, E., Marín, G., & Mpodozis, J. (2020)
Journal of Comparative Neurology, 528: 321– 359. doi: 10.1002/cne.24757.
Can social behaviour drive accessory olfactory bulb asymmetries? Sister species of caviomorph rodents as a case in point.
Fernández-Aburto, P., Delgado, S. E., Sobrero, R., & Mpodozis, J. (2020)
Journal of Anatomy, 236: 612-621. doi: 10.1111/joa.13126.
The nucleus pretectalis principalis: A pretectal structure hidden in the mammalian thalamus.
Deichler, A., Carrasco, D., Gonzalez-Cabrera, C., Letelier, J. C., Marín, G., & Mpodozis, J. (2019)
Journal of Comparative Neurology, 527: 372– 391. doi: 10.1002/cne.24540.
Anatomical specializations related to foraging in the visual system of a nocturnal insectivorous bird, the Band-winged Nightjar (Aves: Caprimulgiformes).
Salazar, J. E., Severin, D., Vega-Zuniga, T., Fernández-Aburto, P., Deichler, A., Sallaberry, M. A., & Mpodozis, J. (2019)
Brain, Behavior and Evolution, 94(suppl 1-4): 27-36. doi: 10.1159/000504162.
“Shepherd’s crook” neurons drive and synchronize the enhancing and suppressive mechanisms of the midbrain stimulus selection network.
Garrido-Charad, F., Vega-Zuniga, T., Gutiérrez-Ibáñez, C., Fernandez, P., López-Jury, L., González-Cabrera, C., Karten, H. J., Luksch, H., & Marín, G. J. (2018)
Proceedings of the National Academy of Sciences, 115(32) E7615-E7623. doi: 10.1073/pnas.1804517115.
Envelope analysis links oscillatory and arrhythmic EEG activities to two types of neuronal synchronization.
Díaz, J., Bassi, A., Coolen, A., Vivaldi, E. A., & Letelier, J. C. (2018)
NeuroImage, 172: 575-585. doi: 10.1016/j.neuroimage.2018.01.063.
Editorial: From ecology to brain development: bridging separate evolutionary paradigms.
Aboitiz, F., Concha, M. L., González-Billault, C., & Mpodozis, J. (2018)
Frontiers in Neuroscience, 12, 447: 1-2. doi: 10.3389/fnins.2018.00447.
Function vs. Structure: The immune system as a case in point.
Mpodozis, J. (2017)
Constructivist Foundations, 13(1): 147–149.
Selective binocular vision loss in two subterranean caviomorph rodents: Spalacopus cyanus and Ctenomys talarum.
Vega-Zuniga, T., Medina, F. S., Marín, G., Letelier, J. C., Palacios, A. G., Němec, P., Schleich, C. E., & Mpodozis, J. (2017)
Scientific Reports, 7, 41704. doi: 10.1038/srep41704.
The centrifugal visual system of a palaeognathous bird, the Chilean Tinamou (Nothoprocta perdicaria).
Krabichler, Q., Vega-Zuniga, T., Carrasco, D., Fernandez, M., Gutiérrez-Ibáñez, C., Marín, G., & Luksch, H. (2017)
Journal of Comparative Neurology, 525: 2514–2534. doi: 10.1002/cne.24195.
Microconnectomics of the pretectum and ventral thalamus in the chicken (Gallus gallus).
Vega-Zuniga, Tomas, Marín, G., González-Cabrera, C., Planitscher, E., Hartmann, A., Marks, V., Mpodozis, J., & Luksch, H. (2016)
Journal of Comparative Neurology, 524: 2208-2229. doi: 10.1002/cne.23941.
Effects of habitat and social complexity on brain size, brain asymmetry and dentate gyrus morphology in two octodontid rodents.
Sobrero, R., Fernández-Aburto, P., Ly-Prieto, Á., Delgado, S. E., Mpodozis, J., & Ebensperger, L. A. (2016)
Brain, Behavior and Evolution, 87: 51-64. doi: 10.1159/000444741.
Bird embryos uncover homology and evolution of the dinosaur ankle.
Ossa-Fuentes, L., Mpodozis, J., & Vargas, A. O. (2015)
Nature Communications, 6, 8902: 1-7. doi: 10.1038/ncomms9902.
Copper enhances cellular and network excitabilities, and improves temporal processing in the rat hippocampus.
Maureira, C., Letelier, J. C., Alvarez, O., Delgado, R., & Vergara, C. (2015)
European Journal of Neuroscience, 42: 3066-3080. doi: 10.1111/ejn.13104.
Early olfactory environment influences social behaviour in adult octodon degus.
Márquez, N., Martínez-Harms, J., Vásquez, R. A., & Mpodozis, J. (2015)
PLoS ONE 10(2): e0118018, 1-12. doi: 10.1371/journal.pone.0118018.
The visual system of a Palaeognathous bird: Visual field, retinal topography and retino‐central connections in the Chilean Tinamou (Nothoprocta perdicaria).
Krabichler, Q., Vega-Zuniga, T., Morales, C., Luksch, H., & Marín, G. J. (2015)
Journal of Comparative Neurology, 523: 226-250. doi: 10.1002/cne.23676.
The isthmic nuclei providing parallel feedback connections to the avian tectum have different neurochemical identities: Expression of glutamatergic and cholinergic markers in the chick (Gallus gallus).
González-Cabrera, C., Garrido-Charad, F., Roth, A., & Marín, G. J. (2015)
Journal of Comparative Neurology, 523: 1341-1358. doi: 10.1002/cne.23739.
Skeletal plasticity in response to embryonic muscular activity underlies the development and evolution of the perching digit of birds.
Francisco Botelho, J., Smith-Paredes, D., Soto-Acuña, S., Mpodozis, J., Palma, V., & Vargas, A. O. (2015)
Scientific Reports, 5, 9840: 1-11. doi: 10.1038/srep09840.
On the hodological criterion for homology.
Faunes, M., Botelho, J. F., Galleguillos, P. A., & Mpodozis, J. (2015)
Frontiers in Neuroscience, 9, 223, 1-7. doi: 10.3389/fnins.2015.00223.
Zebrin II expression in the cerebellum of a Paleognathous bird, the Chilean tinamou (Nothoprocta perdicaria).
Corfield, J. R., Kolominsky, J., Marin, G. J., Craciun, I., Mulvany-Robbins, B. E., Iwaniuk, A. N., & Wylie, D. R. (2015)
Brain, Behavior and Evolution, 85: 94-106. doi: 10.1159/000380810.
Anatomical organization of the visual dorsal ventricular ridge in the chick (Gallus gallus): Layers and columns in the avian pallium.
Ahumada-Galleguillos, P., Fernández, M., Marin, G. J., Letelier, J. C., & Mpodozis, J. (2015)
Journal of Comparative Neurology, 523: 2618-2636. doi: 10.1002/cne.23808.
Morphology, projection pattern, and neurochemical identity of Cajal’s “centrifugal neurons”: The cells of origin of the tectoventrogeniculate pathway in pigeon (Columba livia) and chicken (Gallus gallus).
Vega-Zuniga, Tomas, Mpodozis, J., Karten, H. J., Marín, G., Hain, S., & Luksch, H. (2014)
Journal of Comparative Neurology, 522: 2377-2396. doi:10.1002/cne.23539.
Extracellular recordings reveal absence of magneto sensitive units in the avian optic tectum.
Ramírez, E., Marín, G., Mpodozis, J., & Letelier, J. C. (2014)
Journal of Comparative Physiology A: Neuroethology, Sensory, Neural, and Behavioral Physiology, 200, 983–996. doi: 10.1007/s00359-014-0947-6.
Electroablation: a method for neurectomy and localized tissue injury.
Moya-Díaz, J., Peña, O. A., Sánchez, M., Ureta, D. A., Reynaert, N. G., Anguita-Salinas, C., Marín, G., & Allende, M. L. (2014)
BMC Developmental Biology, 14, 7: 1-13. doi: 10.1186/1471-213X-14-7.
Cognitive ecology in hummingbirds: The role of sexual dimorphism and its anatomical correlates on memory.
González-Gómez, P. L., Madrid-Lopez, N., Salazar, J. E., Suárez, R., Razeto-Barry, P., Mpodozis, J., Bozinovic, F., & Vásquez, R. A. (2014)
PLoS ONE 9(3): e90165, 1-8. doi: 10.1371/journal.pone.0090165.
Does nocturnality drive binocular vision? Octodontine rodents as a case study.
Vega-Zuniga, Tomas, Medina, F. S., Fredes, F., Zuniga, C., Severín, D., Palacios, A. G., Karten, H. J., & Mpodozis, J. (2013)
PLoS ONE 8(12): e84199, 1-12. doi: 10.1371/journal.pone.0084199.
Laminar segregation of GABAergic neurons in the avian nucleus isthmi pars magnocellularis: A retrograde tracer and comparative study.
Faunes, M., Fernández, S., Gutiérrez-Ibáñez, C., Iwaniuk, A. N., Wylie, D. R., Mpodozis, J., Karten, H. J., & Marín, G. (2013)
Journal of Comparative Neurology, 521: 1727-1742. doi: 10.1002/cne.23253.
Attentional capture? Synchronized feedback signals from the isthmi boost retinal signals to higher visual areas.
Marín, G. J., Durán, E., Morales, C., González-Cabrera, C., Sentis, E., Mpodozis, J., & Letelier, J. C. (2012)
Journal of Neuroscience, 32 (3), 1110-1122. doi: 10.1523/JNEUROSCI.4151-11.2012.
Functional implications of species differences in the size and morphology of the isthmo optic nucleus (ION) in birds.
Gutiérrez-Ibáñez, C., Iwaniuk, A. N., Lisney, T. J., Faunes, M., Marín, G. J., & Wylie, D. R. (2012)
PLoS ONE 7(5): e37816, 1-14. doi: 10.1371/journal.pone.0037816.
Bilateral and ipsilateral ascending tectopulvinar pathways in mammals: A study in the squirrel (spermophilus beecheyi).
Fredes, F., Vega-Zuniga, T., Karten, H., & Mpodozis, J. (2012)
Journal of Comparative Neurology, 520: 1800-1818. doi: 10.1002/cne.23014.
Shared and differential traits in the accessory olfactory bulb of caviomorph rodents with particular reference to the semiaquatic capybara.
Suárez, R., Santibáñez, R., Parra, D., Coppi, A. A., Abrahão, L. M. B., Sasahara, T. H. C., & Mpodozis, J. (2011)
Journal of Anatomy, 218: 558-565. doi: 10.1111/j.1469-7580.2011.01357.x.
Deterioration of the Gαo vomeronasal pathway in sexually dimorphic mammals.
Suárez, R., Fernández-Aburto, P., Manger, P. R., & Mpodozis, J. (2011)
PLoS ONE 6(10): e26436, 1-6. doi: 10.1371/journal.pone.0026436
From L’Homme Machine to metabolic closure: Steps towards understanding life.
Letelier, J. C., Cárdenas, M. L., & Cornish-Bowden, A. (2011)
Journal of Theoretical Biology, 286: 100-113. doi: 10.1016/j.jtbi.2011.06.033.
Can red flowers be conspicuous to bees? Bombus dahlbomii and South American temperate forest flowers as a case in point.
Martínez-Harms, J., Palacios, A. G., Márquez, N., Estay, P., Arroyo, M. T. K., & Mpodozis, J. (2010)
Journal of Experimental Biology, 213: 564-571. doi: 10.1242/jeb.037622.
Closure to efficient causation, computability and artificial life.
Luz Cárdenas, M., Letelier, J. C., Gutierrez, C., Cornish-Bowden, A., & Soto-Andrade, J. (2010)
Journal of Theoretical Biology, 263, 1: 79-92. doi: 10.1016/j.jtbi.2009.11.010.
Topographic arrangement of the rotundo‐entopallial projection in the pigeon (Columba livia),
Fredes, F., Tapia, S., Letelier, J. C., Marín, G., & Mpodozis, J. (2010)
Journal of Comparative Neurology, 518: 4342-4361. doi: 10.1002/cne.22460.
Frame-shifts of digit identity in bird evolution and Cyclopamine-treated wings.
Vargas, A. O., & Wagner, G. P. (2009)
Evolution & Development, 11: 163-169. doi: 10.1111/j.1525-142X.2009.00317.x.
Transposition and intermingling of Gαi2 and Gαo afferences into single vomeronasal glomeruli in the Madagascan lesser tenrec Echinops telfairi.
Suárez, R., Villalón, A., Künzle, H., & Mpodozis, J. (2009)
PLoS ONE 4(11): e8005, 1-6. doi: 10.1371/journal.pone.0008005.
Heterogeneities of size and sexual dimorphism between the subdomains of the lateral-innervated accessory olfactory bulb (AOB) of Octodon degus (Rodentia: Hystricognathi).
Suárez, R., & Mpodozis, J. (2009)
Behavioural Brain Research, 198, 2, 306-312. doi: 10.1016/j.bbr.2008.11.009.
Novel, continuous visual motion induces c-fos expression in the avian optokinetic nuclei and optic tectum.
Rojas, X., Marín, G., & Wallman, J. (2009)
Neuroscience, 160, 2, 540-554. doi: 10.1016/j.neuroscience.2009.02.018.
Patterns of skull variation of brachydelphis (Cetacea, Odontoceti) from the neogene of the southeastern pacific.
Gutstein, C. S., Cozzuol, M. A., Vargas, A. O., Suárez, M. E., Schultz, C. L., & Rubilar-Rogers, D. (2009)
Journal of Mammalogy, 90, 2, 504–519. doi: 10.1644/07-MAMM-A-081.1
On the nature of thumbs.
Wagner, G. P., & Vargas, A. O. (2008)
Genome Biology, 9, 213, 1-3. doi: 10.1186/gb-2008-9-3-213.
The evolution of HoxD-11 expression in the bird wing: Insights from Alligator mississippiensis.
Vargas, A. O., Kohlsdorf, T., Fallon, J. F., VandenBrooks, J., & Wagner, G. P. (2008)
PLoS ONE 3(10): e3325, 1-7. doi: 10.1371/journal.pone.0003325.
Differences of morphological and ecological characters among lineages of Chilean Rhinocryptidae in relation an sister lineage of Furnariidae.
Correa Rueda, A., Mpodozis, J., & Sallaberry, M. (2008)
Nature Precedings. doi: 10.1038/npre.2008.1606.1.
Speciation of chilean Rhinocryptidae (Avian) based on their behaviour.
Correa, A., & Mpodozis, J. (2008)
Nature Precedings. doi: 10.1038/npre.2008.1597.2.
A cholinergic gating mechanism controlled by competitive interactions in the optic tectum of the pigeon.
Marín, G., Salas, C., Sentis, E., Rojas, X., Letelier, J. C., & Mpodozis, J. (2007)
Journal of Neuroscience, 27, (30), 8112-8121. doi: 10.1523/JNEUROSCI.1420-07.2007.
Amplitude modulation patterns of local field potentials reveal asynchronous neuronal populations.
Díaz, J., Razeto-Barry, P., Letelier, J. C., Caprio, J., & Bacigalupo, J. (2007)
Journal of Neuroscience, 27, (34), 9238-9245. doi: 10.1523/JNEUROSCI.4512-06.2007.
Beyond reductionism: Metabolic circularity as a guiding vision for a real biology of systems.
Cornish-Bowden, A., Cárdenas, M. L., Letelier, J. C., & Soto-Andrade, J. (2007)
Proteomics, 7: 839-845. doi: 10.1002/pmic.200600431.
Organizational invariance and metabolic closure: Analysis in terms of (M, R) systems.
Letelier, J. C., Soto-Andrade, J., Guíñez Abarzúa, F., Cornish-Bowden, A., & Luz Cárdenas, M. (2006)
Journal of Theoretical Biology, 238, 4, 949-961. doi: 10.1016/j.jtbi.2005.07.007.
Influencing and interpreting visual input: The role of a visual feedback system.
Gruberg, E., Dudkin, E., Wang, Y., Marín, G., Salas, C., Sentis, E., Letelier, J., Mpodozis, J., Malpeli, J., Cui, H., Ma, R., Northmore, D., & Udin, S. (2006)
Journal of Neuroscience, 26, (41), 10368-10371. doi: 10.1523/JNEUROSCI.3288-06.2006.
The digits of the wing of birds are 1, 2, and 3. A review.
Vargas, A. O., & Fallon, J. F. (2005)
Journal of Experimental Zoology - Part B: Molecular and Developmental Evolution, 304B: 206-219. doi:10.1002/jez.b.21051.
Birds have dinosaur wings: The molecular evidence.
Vargas, A. O., & Fallon, J. F. (2005)
Journal of Experimental Zoology - Part B: Molecular and Developmental Evolution, 304B: 86-90. doi:10.1002/jez.b.21023.
How ancient is the adult swimming capacity in the lineage leading to Euchordates?
Vargas, A. O., & Aboitiz, F. (2005)
Evolution & Development, 7: 171-174. doi: 10.1111/j.1525-142X.2005.05029.x.
Oscillatory bursts in the optic tectum of birds represent re-entrant signals from the nucleus isthmi pars parvocellularis.
Marín, G., Mpdozis, J., Sentis, E., Ossandón, T., & Letelier, J. C. (2005)
Journal of Neuroscience, 25, (30), 7081-7089. doi: 10.1523/JNEUROSCI.1379-05.2005.
Beyond selection. Más allá de la selección.
Vargas, A. O. (2005)
Revista Chilena de Historia Natural, 78, 4, 739-752. doi: 10.4067/S0716-078X2005000400012.
Undergraduate teaching of evolution in Chile: More than natural selection.
Ahumada, P., Astorga, G., Bull-Hereñu, K., Cabrera, R., Caputo, L., Carmona, C., Cecchi, M. C., Cortés, N., Cortez, S., Dorador, C., Fredes, F., Guerrero-Bosagna, C., Henny, P., Libedinsky, C., Marcelo, W., Márquez, I. N., Martínez, J., Medina, M., Parra, D., Pohl, N., Potttock, H., Ossandón, T., Razeto, P., Rubilar, D., Salas, C., Salinas, F., Sepúlveda, E., Soto, C., Suárez, R., Vargas, A., Vega-Zúñiga, T., Velásquez, N., Vidal., J., Villagra., C.A., & Villavicencio, C. P. (2005)
Revista Chilena de Historia Natural, 78, 1, 169-170. doi: 10.4067/S0716-078X2005000100013.
The mapping of the visual field onto the dorso-lateral tectum of the pigeon (Columba livia) and its relations with retinal specializations.
Letelier, J. C., Marin, G., Sentis, E., Tenreiro, A., Fredes, F., & Mpodozis, J. (2004)
Journal of Neuroscience Methods, 132, 2, 161-168. doi: 10.1016/j.jneumeth.2003.09.007.
Understanding the parts in terms of the whole.
Cornish-Bowden, A., Cárdenas, M. L., Letelier, J. C., Soto-Andrade, J., & Abarzúa, F. G. (2004)
Biology of the Cell, 96, 9, 713-717. doi: 10.1016/j.biolcel.2004.06.006.
Answering Cuvier: Notes on the systemic/historic nature of living beings.
Cecchi, M. C., Vargas, A., Villagra, C. A., & Mpodozis, J. (2004)
Cybernetics & Human Knowing, 11, 4, 2004, 11-19(9).
Spatial organization of the pigeon tectorotundal pathway: An interdigitating topographic arrangement.
Marín, G., Letelier, J. C., Henny, P., Sentis, E., Farfán, G., Fredes, F., Pohl, N., Karten, H., & Mpodozis, J. (2003)
Journal of Comparative Neurology, 458: 361-380. doi: 10.1002/cne.10591.
Autopoietic and (M,R) systems.
Letelier, J. C., Marín, G., & Mpodozis, J. (2003)
Journal of Theoretical Biology, 222, 2, 261-272. doi: 10.1016/S0022-5193(03)00034-1.
Emplumando dinosaurios: La transición evolutiva de terópodos a aves.
Chiappe, L. M., & Vargas, A. (2003)
Hornero, 18, (1), 1-11.
Metabolic networks from (M, R) systems and autopoiesis perspective.
Zaretzky, A. N., & Letelier, J. C. (2002)
Journal of Biological Systems, 10, 03, 265-280. doi: 10.1142/S0218339002000573.
Los derroteros científicos de Francisco Varela (1946-2001).
Letelier, J. C. (2001)
Biological Research, 34, (2), 7-13. doi: 10.4067/S0716-97602001000200002.
A simple method to microinject solid neural tracers into deep structures of the brain.
Marín, G., Henny, P., Letelier, J. C., Sentis, E., Karten, H., Mrosko, B., & Mpodozis, J. (2001)
Journal of Neuroscience Methods, 106, 2, 121-129. doi: 10.1016/S0165-0270(01)00332-6.
El ¿delito? de Aristóteles.
Cecchi, M. C., Guerrero-Bosagna, C., & Mpodozis, J. (2001)
Revista Chilena de Historia Natural, 74, (3), 507-514. doi: 10.4067/S0716-078X2001000300001.
The origin of species by means of natural drift.
Maturana, H., & Mpodozis, J. (2000)
Revista Chilena de Historia Natural, 73, (2), 261-310. doi: 10.4067/S0716-078X2000000200005.
Spike sorting based on discrete wavelet transform coefficients.
Letelier, J. C., & Weber, P. P. (2000)
Journal of Neuroscience Methods, 101, 2, 93-106. doi: 10.1016/S0165-0270(00)00250-8.
Spatiotemporal profile of synaptic activation produced by the electrical and visual stimulation of retinal inputs to the optic tectum: A current source density analysis in the pigeon (Columba livia).
Letelier, J. C., Mpodozis, J., Marin, G., Morales, D., Rozas, C., Madrid, C., & Velasco, M. (2000)
European Journal of Neuroscience, 12: 47-57. doi: 10.1046/j.1460-9568.2000.00872.x.
Evolution of arm size in theropod dinosaurs: a developmental hypothesis.
Vargas, A. O. (1999)
Noticiario Mensual del Museo Nacional de Historia Natural, Chile, 338: 16-19.
Reversible mechanical fixation of eye position in awake head-restrained pigeons (Columba livia).
Letelier, J. C., Mpodozis, J., Marin, G., Morales, D., Henny, P., Madrid, C., & Velasco, M. (1999)
Journal of Neuroscience Methods, 91, 1–2, 67-71. doi: 10.1016/S0165-0270(99)00073-4.
Two distinct populations of tectal neurons have unique connections within the retinotectorotundal pathway of the pigeon (Columba livia).
Karten, H. J., Cox, K., & Mpodozis, J. (1997)
Journal of Comparative Neurology, 387: 449-465. doi: 10.1002/(SICI)1096-9861(19971027)387:3<449::AID-CNE10>3.0.CO;2-G.
GABAergic inputs to the nucleus rotundus (pulvinar inferior) of the pigeon (Columba livia).
Mpodozis, J., Cox, K., Shimizu, T., Bischof, H. J., Woodson, W., & Karten, H. J. (1996)
Journal of Comparative Neurology, 374: 204-222. doi: 10.1002/(SICI)1096-9861(19961014)374:2<204::AID-CNE4>3.0.CO;2-6.
RETINET: A Neural Network for the Psychophysics of Color Vision.
Glaria-Bengoechea, A., Mpodozis Marin, J., Jonas-Zuñiga, R., & Ballesteros Leiva, A. (1996)
Proceedings of International Conference on Neural Networks (ICNN’96), Washington, DC, USA, 1996, pp. 724-727 vol.2. doi: 10.1109/ICNN.1996.548985.
Linear analysis of auto-organization in Hebbian neural networks.
Letelier, J. C., & Mpodozis, J. (1995)
Biological Research, 28, (1), 97-104.
Conduction velocity groups in the retino-tectal and retino-thalamic visual pathways of the pigeon (Columba livia).
Mpodozis, J., Letelier, J. C., Concha, M. L., & Maturana, H. (1995)
International Journal of Neuroscience, 81:3-4, 123-136. doi: 10.3109/002074595090153.04
Brain, language and the origin of human mental functions.
Maturana, H., Mpodozis, J., & Letelier, J. C. (1995)
Biological Research, 28, (1), 15-26.
The retinal targets of centrifugal neurons and the retinal neurons projecting to the accessory optic system.
Nickla, D., Gottlieb, M., Marin, G., Rojas, X., Britto, L., & Wallman, J. (1994)
Visual Neuroscience, 11, (2), 401-409. doi: 10.1017/S0952523800001747.
Effects of retinal lesions upon the distribution of nicotinic acetylcholine receptor subunits in the chick visual system.
Britto, L. R.G., Torrao, A. S., Hamassaki‐Britto, D. E., Mpodozis, J., Keyser, K. T., Lindstrom, J. M., & Karten, H. J. (1994)
Journal of Comparative Neurology, 350: 473-484. doi: 10.1002/cne.903500311.
Alternativas de automatización para el guiado autónomo de vehículos cargadores frontales en una mina subterránea.
Córdova, F., Doggenweiler, C., Maturana, H., Mpodozis, J., Letelier, J. C., & Moyano, A. (1993)
Automática e Innovación, 2: 67–63.
Saccade-related responses of centrifugal neurons projecting to the chicken retina.
Marin, G., Letelier, J. C., & Wallman, J. (1990)
Experimental Brain Research, 82, 263–270. doi: 10.1007/BF00231246.
Lenguaje y realidad: el origen de lo humano.
Maturana, H. (1989)
Archivos de Biología y Medicina Experimentales, 22: 77-81.
Morphic resonance in silicon chips: An experimental test of the hypothesis of formative causation.
Varela, F., & Letelier, J. C. (1988)
Skeptical Inquirer, 12, 3: 298–300.
A quantitative assessment of the dependency of the visual temporal frame upon the cortical rhythm.
Gho, M., & Varela, F. J. (1988)
Journal of Physiology (Paris), 83, (2):95-101.
Frontal and lateral visual system in birds.
Maldonado, P. E., Maturana, H., & Varela, F. (1988)
Brain, Behavior and Evolution, 32: 57–62. doi: 10.1159/000116532.
Perception: Behavioral configuration of the object.
Maturana, H., & Mpodozis, J. (1987)
Archivos de Biología y Medicina Experimentales, 20, (3-4), 319–324.
The organ of form: towards a theory of biological shape.
Varela, F. J., & Frenk, S. (1987)
Journal of Social and Biological Structures, 10, 1, 73-83. doi: 10.1016/0140-1750(87)90035-2.
Exact results for the average dynamic behavior of some non-linear neural networks.
Rössler, J., & Varela, F. J. (1987)
Biological Cybernetics, 57, 249–256. doi: 10.1007/BF00338818.
Cytoarchitecture of the avian ventral lateral geniculate nucleus.
Guiloff, G. D., Maturana, H. R., & Varela, F. J. (1987)
Journal of Comparative Neurology, 264: 509-526. doi: 10.1002/cne.902640406.
Self-reference and fixed points: A discussion and an extension of Lawvere’s Theorem.
Soto-Andrade, J., & Varela, F. J. (1984)
Acta Applicandae Mathematica 2, 1–19. doi: 10.1007/BF01405490.
Why the cortical magnification factor in rhesus is isotropic.
Letelier, J. C., & Varela, F. (1984)
Vision Research, 24, 9, Pages 1091-1095. doi: 10.1016/0042-6989(84)90087-7.
Scientists in Pinochet ’s Chile.
Cordero, P., Garreton, M. A., Giannini, H., Goic, A., Izquierdo, L., Latorre, R., Minguell, J., Ureta, T., Varela, F., & Vivaldi, E. (1984)
Nature, 308, 310. doi: 10.1038/308310a0.
Regional specialization of the quail retina: Ganglion cell density and oil droplet distribution.
Budnik, V., Mpodozis, J., Varela, F. J., & Maturana, H. R. (1984)
Neuroscience Letters, 51, 1, 145-150. doi: 10.1016/0304-3940(84)9027.6-3.
The neurophysiology of avian color vision.
Maturana, H., Varela, F., Marin, G., & Letelier, J. C. (1983)
Archivos de Biología y Medicina Experimentales, 16: 291–303.
What is it to see?
Maturana, H. R. (1983)
Archivos de Biología y Medicina Experimentales, 16(3-4): 255-269.
La corteza bien temperada: Análisis harmónico de campos receptivos visuales.
Varela, F. (1982)
Archivos de Biología y Medicina Experimentales, 15: 297–303.
Las álgebras de Boole y la noción de distinción.
Varela, F. (1982)
Notas de la Sociedad de Matemática de Chile, 1: 39-53.
Color-opponent responses in the avian lateral geniculate: A study in the quail (Coturnix coturnix japonica).
Maturana, H. R, & Varela, F. J. (1982)
Brain Research, 247, 2, 227-241. doi: 10.1016/0006-8993(82)91248-3.
Perceptual framing and cortical alpha rhythm.
Varela, F. J., Toro, A., John, E. R., & Schwartz, E. L. (1981)
Neuropsychologia, 19, 5, 675-686. doi: 10.1016/0028-3932(81)90005-1.
Size constancy and accommodation.
Maturana, H. R., & Varela, F. J. (1981)
Perception, 10, (6), 707–709. doi: 10.1068/p100707.
The quest for the intelligence of intelligence.
Maturana, H. R., & Guiloff, G. D. (1980)
Journal of Social and Biological Structures, 3, 2, 135-148. doi: 10.1016/0140-1750(80)90006-8.
Class I retinal ganglion cells in the toad, Bufo spinulosus.
Guiloff, G. D. (1980)
Vision Research, 20, 6, 549-551. doi: 10.1016/0042-6989(80)90131-5.
The organization of the living : A theory of the living organization.
Maturana, H. R. (1975)
International Journal of Man-Machine Studies, 7, 3, 313-332. doi: 10.1016/S0020-7373(75)80015-0.
A calculus for self-reference.
Varela, F. J. (1975)
International Journal of General Systems, 2, 1, 5-24. doi: 10.1080/03081077508960828.
Autopoiesis: The organization of living systems, its characterization and a model.
Varela, F. G., Maturana, H. R., & Uribe, R. (1974)
Biosystems, 5, 4, 187-196. doi: 10.1016/0303-2647(74)90031-8.
Mechanism and biological explanation.
Varela, F., & Maturana, H. (1972)
Philosophy of Science, 39, 3, 378-382. doi: 10.1086/288458.
Size constancy and the problem of perceptual spaces.
Maturana, H. R., Varela, F. G., & Frenk, S. G. (1972)
Cognition, 1, 1, 97-104. doi: 10.1016/0010-0277(72)90047-9.
The vertebrate and the (insect) compound eye in evolutionary perspective.
Varela, F. G. (1971)
Vision Research, 11, 3, 201-209. doi: 10.1016/0042-6989(71)90040-X.
Oil droplet distribution and colour discrimination in the pigeon.
Bloch, S., & Maturana, H. R. (1971)
Nature New Biology 234, 284–285. doi: 10.1038/newbio234284a0.
Are colour oil droplets the basis of the pigeon’s chromatic space?
Bloch, S., & Martinoya, C. (1971)
Vision Research, 11, 3, 411-418. doi: 10.1016/0042-6989(71)90054-X.
Biology of cognition.
Maturana, H. (1970)
Biological Computer Laboratory Research Report BCL 9.0. Urbana IL: University of Illinois, 1970.
Time courses of excitation and inhibition in retinal ganglion cells.
Varela, F. G., & Maturana, H. R. (1970)
Experimental Neurology, 26, 1, 53-59. doi: 10.1016/0014-4886(70)90087-7.
A biological theory of relativistic colour coding in the primate retina.
Maturana, H., Uribe, G., & Frenk, S. (1968)
Archivos de Biología y Medicina Experimentales, 1: 1-30.
Synaptic connections of the centrifugal fibers in the pigeon retina.
Maturana, H. R., & Frenk, S.
Science, 150, 3694, 359-361. doi: 10.1126/science.150.3694.359.
Octopus optic responses.
Boycott, B. B., Lettvin, J. Y., Maturana, H. R., & Wall, P. D. (1965)
Experimental Neurology, 12, 3, 247-256. doi: 10.1016/0014-4886(65)90070-1.
Especificidad vs ambigüedad en la retina de los vertebrados.
Maturana, H. (1964)
Biologica, fascículo XXXVI, 69-96.
Directional movement and horizontal edge detectors in the pigeon retina.
Maturana, H., & Frenk, S. (1963)
Science, 142, 3594, 977-979. doi: 10.1126/science.142.3594.977.